Why this paper matters on a busy ICU day
Non-invasive respiratory support (NIRS) is now everywhere: ED, step-down, wards, and ICU. We use HFNT, CPAP, BiPAP, facemasks, helmets — often driven by habit or availability more than physiology.
Rezoagli and colleagues offer a practical, physiology-heavy roadmap for how to choose a modality, how to set it up, and when to walk away and intubate. This is the kind of paper that can clean up practice variation across an entire service.
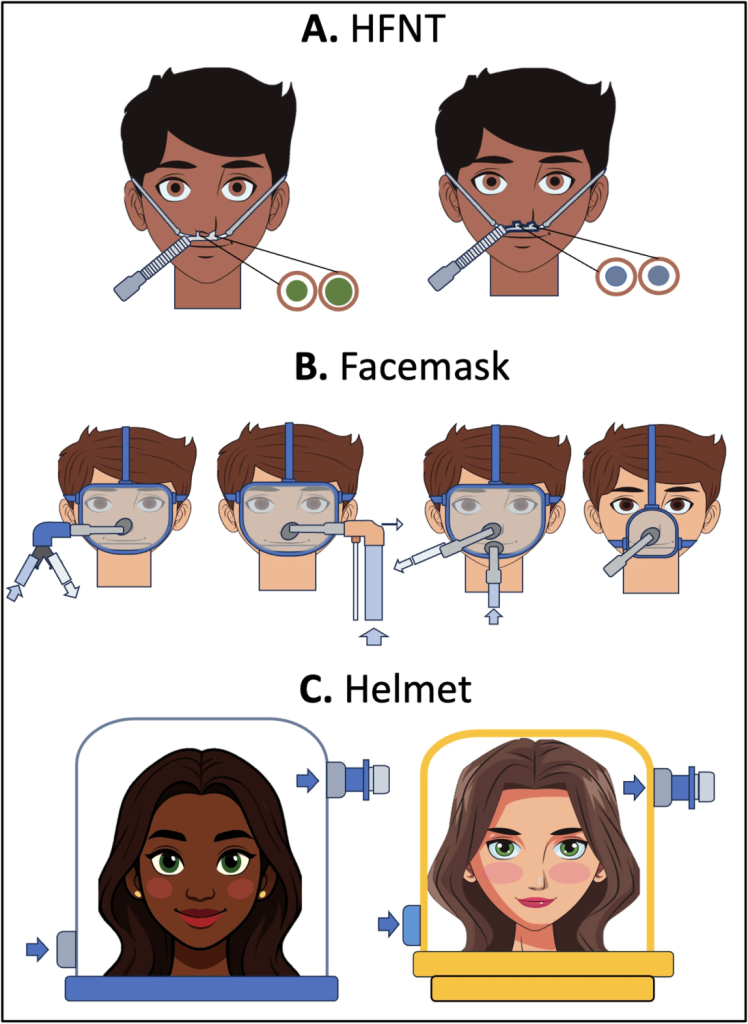
1. Big picture: what the article actually does
The review walks through three pillars:
Physiology & technical setup
- High flow nasal therapy (HFNT)
- CPAP
- BiPAP (pressure-support NIV)
- Facemasks vs helmets
- Humidification and temperature
Clinical indications by syndrome
- Cardiogenic pulmonary edema (CPE)
- De novo acute hypoxemic respiratory failure (AHRF, including ARDS and pneumonia)
- Hypercapnic failure in AECOPD
- Post-extubation support
- Obesity hypoventilation and cystic fibrosis
Monitoring and when to stop
- How to detect failure early and avoid P-SILI from “too much effort for too long” on NIRS.
The strength of the paper is that it links physiology to concrete bedside choices: interface, flow, PEEP, pressure support, and monitoring.
2. HFNT – the workhorse for hypoxemic patients
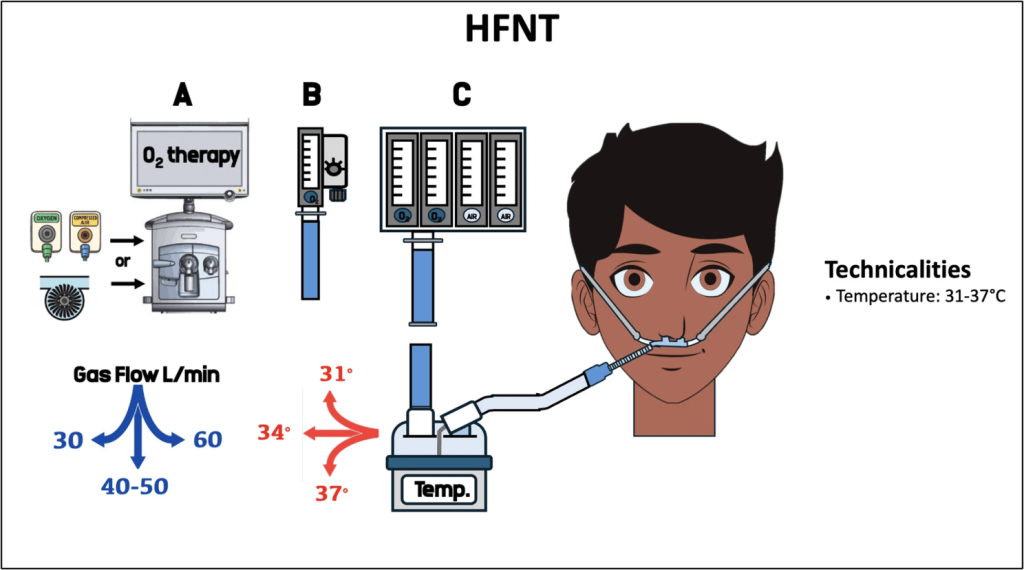
Key physiological points
- HFNT delivers heated, humidified gas at 30–60 L/min via nasal cannula, often starting at 60 L/min in AHRF.
- At higher flows, it:
- Provides low-level PEEP (~1–4 cmH₂O), especially with the mouth closed.
- Washes out upper airway dead space, improving ventilatory efficiency and allowing lower minute ventilation for the same PaCO₂.
- Increases end-expiratory lung volume and can help prevent alveolar collapse in AHRF.
Practical nuances
- Flows
- AHRF: start high (≈60 L/min) to maximize EELV and oxygenation.
- Hypercapnic COPD: 30–40 L/min often sufficient; higher flows don’t add much for CO₂ clearance and may increase discomfort.
- Temperature
- Absolute humidity is best at higher temperatures, but patients are often more comfortable at ~31°C vs 37°C — start cooler and titrate to comfort.
- Asymmetrical cannulas
- Newer “asymmetrical” HFNT cannulas may enhance CO₂ washout and reduce work of breathing by exploiting pressure differences between nares, though outcome data remain limited.
Clinical implication for you
HFNT is positioned as the first-line modality for de novo AHRF, including many ARDS and pneumonia patients who are not intubated yet. It offers a good balance of support, comfort, and ease of use — as long as you monitor closely for failure.
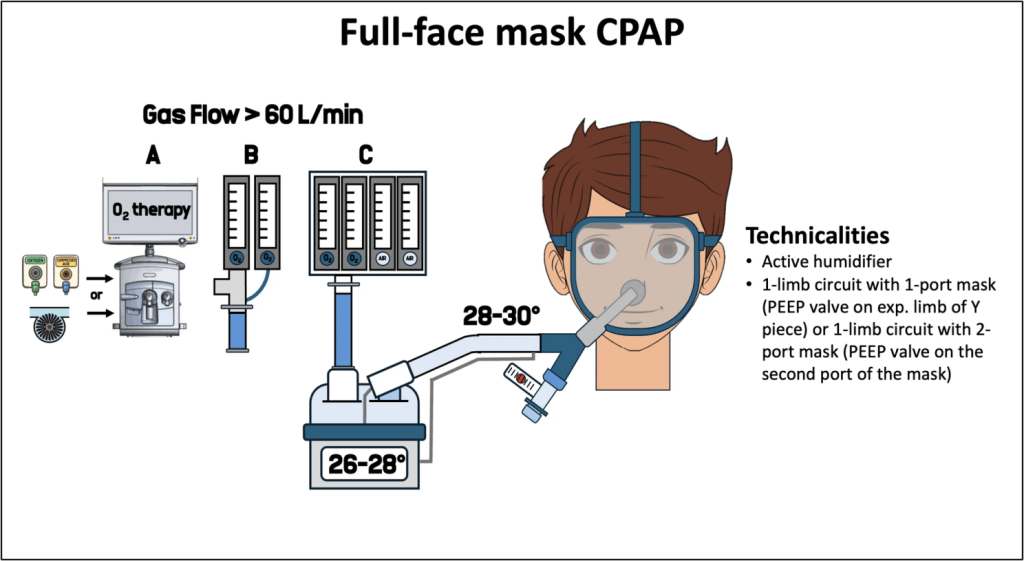
3. CPAP – simple PEEP with powerful heart–lung effects
What CPAP actually does
- Provides constant positive pressure through the respiratory cycle (PEEP).
- In injured lungs, this:
- Increases end-expiratory lung volume and recruits alveoli.
- Moves ventilation onto the more linear part of the pressure–volume curve.
- Improves V/Q matching and oxygenation.
- Hemodynamically, CPAP:
- Reduces venous return and RV preload, which can reduce pulmonary congestion.
- Lowers LV afterload by reducing negative intrathoracic swings and transmural LV pressure — particularly beneficial in LV failure.
Delivery options
- ICU, transport, or home ventilators (CPAP mode or PSV with PS=0).
- “Frugal” systems: Venturi devices, Boussignac or similar valves, and oxygen-saving CPAP setups for resource-limited settings or pre-hospital care.
- Helmet CPAP requires high flows (≥60 L/min) to avoid CO₂ rebreathing and maintain stable PEEP.
Where CPAP shines
- Cardiogenic pulmonary edema (CPE)
- NIV (CPAP or BiPAP) is first-line alongside medical therapy in CPE without shock or urgent PCI needs.
- No clear winner between CPAP and BiPAP; choice should focus on leaks, CO₂ clearance, and tolerance.
- Helmet CPAP may provide faster short-term respiratory and hemodynamic improvement vs HFNT.
4. BiPAP – unloading the muscles and clearing CO₂
Core physiology
- BiPAP = PEEP + inspiratory pressure (PS).
- Adds:
- Higher minute ventilation and CO₂ clearance.
- Reduced inspiratory effort and work of breathing.
- In heart failure, BiPAP combines the CPAP benefits with active ventilatory support.
The double-edged sword in AHRF
- If BiPAP fails to reduce inspiratory effort in hypoxemic AHRF, transpulmonary pressures and tidal volumes can rise, worsening P-SILI.
- This is why careful monitoring of effort, tidal volume (if measurable), and clinical trajectory is crucial.
In COPD and hyperinflation
- COPD patients often have dynamic hyperinflation and intrinsic PEEP (PEEPi).
- BiPAP with external PEEP ≈ 5–8 cmH₂O can act as a “PEEP absorber” by holding small airways open and reducing the inspiratory threshold load.
- In AECOPD with acidosis, BiPAP via facemask is clearly first-line — robust data show reductions in intubation and mortality compared with conventional oxygen.
5. Interfaces, circuits, and humidification – the “plumbing” that matters
Facemask vs helmet

- Facemask
- Oro-nasal or full-face masks; similar inspiratory effort and CO₂ clearance overall.
- Common issues: leaks, skin breakdown over the nasal bridge, limited ability to eat and talk, claustrophobia.
- Helmet
- Transparent hood with neck seal, large internal volume (~18 L).
- Advantages: less facial trauma, better tolerance for prolonged use, easier communication and mobilization, high PEEP capability.
- Challenges: noise, potential for CO₂ rebreathing (needs high flow and proper circuit configuration), more complex line management around the neck.
- Newer helmet BiPAP designs have reduced internal volume and compliance, improving pressurization and synchrony.
Humidification
- For CPAP/BiPAP:
- Active heated humidification generally preferred, especially in hypercapnic patients, because it improves alveolar ventilation and CO₂ clearance and reduces WOB.
- HMEs add dead space and resistance; they are simple but less physiologically favorable.
- Target gas temperature around 26–28°C at the humidifier, aiming for ~30°C at the interface, balanced against patient comfort and condensation risk.
6. Indications by clinical syndrome – how the authors would “triage” support
6.1 Cardiogenic pulmonary edema (CPE)
- NIV (CPAP or BiPAP) + medical therapy is first-line in CPE without cardiogenic shock.
- No clear superiority of CPAP vs BiPAP; choose based on tolerance and need for CO₂ clearance.
- HFNT is non-inferior to facemask BiPAP for short-term outcomes and may be useful for prolonged support or in patients who cannot tolerate CPAP/BiPAP.
- Helmet CPAP may provide faster hemodynamic and respiratory improvement than HFNT.
6.2 De novo acute hypoxemic respiratory failure (AHRF)
- Includes ARDS, unilateral pneumonia, aspiration, contusion, septic shock without chronic lung disease.
- HFNT:
- Supported by FLORALI and subsequent trials as first-line therapy, with lower intubation rates in more hypoxemic subgroups and better comfort than facemask NIV in many settings.
- CPAP/BiPAP:
- Evidence is mixed; some network meta-analyses show mortality and intubation benefits vs conventional oxygen, others are neutral when you exclude CPE/COPD.
- For COVID-19 AHRF, CPAP reduced intubation compared with conventional oxygen, whereas HFNT did not clearly outperform oxygen in the RECOVERY-RS trial.
- The review ultimately endorses HFNT as first-line in non-cardiogenic AHRF in the ICU, with CPAP/BiPAP reserved for selected cases and used under tight monitoring.
6.3 AECOPD with hypercapnic respiratory failure
- Strong RCT and meta-analytic data: BiPAP via facemask reduces intubation and mortality.
- Higher-intensity BiPAP (higher PS targeting significant pH and PaCO₂ improvement) further decreases intubation risk compared with low-intensity strategies.
- HFNT offers physiological benefits but has not matched BiPAP in preventing invasive ventilation; non-inferiority was not consistently demonstrated except in some mixed populations.
- HFNT is best seen as adjunct/bridge when BiPAP is poorly tolerated or during breaks.
6.4 Post-extubation support
- High-risk patients (age, comorbidity, prolonged ventilation):
- NIV (often alternating with HFNT) is superior to HFNT alone for preventing reintubation.
- Guidelines now suggest NIV over HFNT in this group.
- Lower-risk patients:
- HFNT is favored over conventional oxygen, although evidence quality is lower.
- Non-invasive support after extubation should be used early and for prolonged periods (≥12 h in first 24 h) to preserve lung recruitment and reduce muscle load.
6.5 OHS and cystic fibrosis
- Acute OHS decompensation
- BiPAP is the preferred acute modality: maintains upper airway patency, unloads respiratory muscles, improves alveolar ventilation.
- Full-face masks are more effective than nasal masks in the acute setting.
- Cystic fibrosis exacerbations
- BiPAP is widely used to support fatigued respiratory muscles and improve gas exchange; data are mostly observational but consistent.
- HFNT can be a useful adjunct, reducing respiratory rate and minute ventilation.
7. Monitoring, failure, and avoiding P-SILI
The authors are very clear: non-invasive does not mean non-dangerous.
Key monitoring concepts:
- Watch the trajectory
- Rapid improvement in CPE and AECOPD is expected; persistent acidosis or hypercapnia under optimized settings signals NIV failure.
- In AHRF, the risk is P-SILI
- Strong spontaneous efforts → large negative pleural swings → high transpulmonary pressures and pendelluft → progression of lung injury.
- Parameters to track
- Respiratory rate, dyspnea, accessory muscle use.
- Gas exchange trends (PaO₂/FiO₂, PaCO₂, pH).
- Simple indices and clinical scores for NIV failure.
- Tidal volume (where measurable) to avoid large VT.
- Bedside lung ultrasound and, where available, advanced tools such as esophageal pressure monitoring or electrical impedance tomography to assess effort, regional ventilation, and pendelluft.
The take-home is: non-invasive support must sit inside a tight monitoring framework with a low threshold for intubation when the trajectory is wrong.
Bottom line for your practice
- HFNT is now the default first-line for de novo hypoxemic AHRF in the ICU, provided you monitor closely and have a clear plan for escalation.
- BiPAP via facemask remains the undisputed standard for AECOPD with respiratory acidosis and is central in OHS exacerbations and CF crises.
- CPAP (facemask or helmet) is a powerful hemodynamic tool in CPE and a viable option in selected hypoxemic patients, especially in COVID-19 pneumonia.
- The choice of interface and circuit (helmet vs facemask, single vs double limb, HME vs heated humidifier) is not cosmetic; it directly affects CO₂ clearance, comfort, and success.
- Above all: monitor relentlessly. Non-invasive support can prevent intubation, but if misused or prolonged in a failing patient, it can worsen lung injury.

Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.

