1. Why This Article Matters
Invasive mechanical ventilation is the cornerstone of critical care. Yet, one major risk remains overlooked in daily practice:
Ventilator exhaust can carry infectious aerosols, biofilm fragments, volatile anesthetics, and VOCs into the ICU environment — posing real risks for patients and staff.
This review synthesizes decades of microbiology, aerosol physics, engineering controls, and global outbreak investigations to answer a question we rarely consider:
What is actually coming out of the ventilator exhaust — and how dangerous is it?
Shi et al. deliver one of the most comprehensive analyses to date, showing that ventilator exhaust represents a patient → device → environment → staff exposure chain, and they propose a modern, tiered prevention strategy designed for real-world ICU workflows.
This paper is exceptionally relevant for RTs, intensivists, infection-prevention teams, and hospital leadership.

2. The Article in 5 Lines
- Ventilator exhaust contains bioaerosols, pathogens, volatile anesthetics, and VOCs, all of which can enter the clinical environment.
- Aerosol dispersion occurs through an under-recognized transmission chain: patient → circuit → exhaust → ICU air → staff/patients.
- High-risk amplifiers include suctioning, nebulization, bronchoscopies, inhaled volatile sedation, and circuit condensation.
- Mitigation options exist (HMEF, HEPA, directed discharge, chemical inactivation) but vary widely in evidence, complexity, and safety.
- Shi et al. propose a three-tier prevention framework aligned with patient condition, infection status, and ICU engineering controls.

3. Key Insights You Need to Know
1️⃣ Ventilator exhaust is not just “air” — it is a complex aerosol stream
The exhalation valve releases:
- bioaerosols containing bacteria & viruses
- evaporated or aerosolized condensate
- volatile anesthetic waste gases
- VOCs such as toluene and ethylbenzene
Particle sizes <5 μm stay airborne for hours and travel meters — contaminating staff breathing zones and increasing cross-infection risk.
2️⃣ Biofilm fragments from the ETT can aerosolize and spread via exhaust
The review highlights a biofilm-driven dissemination mechanism:
- biofilms form on the ETT
- expiratory airflow shears them into the circuit
- fragments travel into the ventilator and out through the exhaust
- they can be re-inhaled by the same patient or others This mechanism is visually depicted in Figure 1 (page 4).
This is a major contributor to VAP pathogenesis — far beyond classic microaspiration.
3️⃣ Real-world outbreaks confirm exhaust-associated infections
Table 1 (pages 5–10) documents cases dating back to 1952 involving:
- Streptococcus pyogenes
- Pseudomonas aeruginosa
- MDR Klebsiella pneumoniae
- Vancomycin-resistant enterococci
- Acinetobacter baumannii
In multiple events, the ventilator exhaust was the direct source of airborne spread.
4️⃣ High-risk procedures and therapies massively amplify aerosol release
The review identifies strong emission generators:
- suctioning
- coughing
- nebulization (especially bronchodilators, corticosteroids, antibiotics)
- inhaled volatile anesthetics (waste gases)
Notably: Even quiet breathing from “super-spreaders” can emit submicron aerosols (<1 μm).
Figure 3 (page 6) maps the contamination zones clearly.

5️⃣ Existing filtration solutions differ dramatically in performance
From Table 2 (page 11):
- HME/HMEF = balanced effectiveness, low cost, easy to implement
- HEPA = highest filtering efficiency but increased resistance & maintenance burden
- Chemical inactivation devices = low cost but limited evidence
- Directed exhaust discharge = reduces room contamination but requires infrastructure
- Anesthetic scavenging systems (WAGS) = promising but largely theoretical in ICU ventilation
The radar plot on page 12 shows performance differences across 6 dimensions.
6️⃣ ICU ventilation engineering plays a major protective role
Air-change rate and directional airflow influence:
- particle concentration
- residence time
- cross-zone contamination
Key benchmarks:
- ≥6 ACH for routine care
- ≥12 ACH for AGPs
- portable HEPA units increase “effective ACH”
CFD simulations (pages 12–13) show how room airflow patterns can create contamination hotspots invisible to staff.
7️⃣ Nebulizer-generated “fugitive aerosols” escape into the exhaust
These combine with pathogens from pulmonary infection, creating a compounded aerosol burden. Expiratory limb filtration is essential.
8️⃣ Inhaled anesthetics introduce a second hazard: chronic staff exposure
Volatile agents are exhaled unchanged and released into the room unless scavenged. Chronic exposure is linked to:
- oxidative stress
- cytokine dysregulation
- multi-organ injury
- possible malignancy
ICU providers rarely consider this risk — yet it is real.
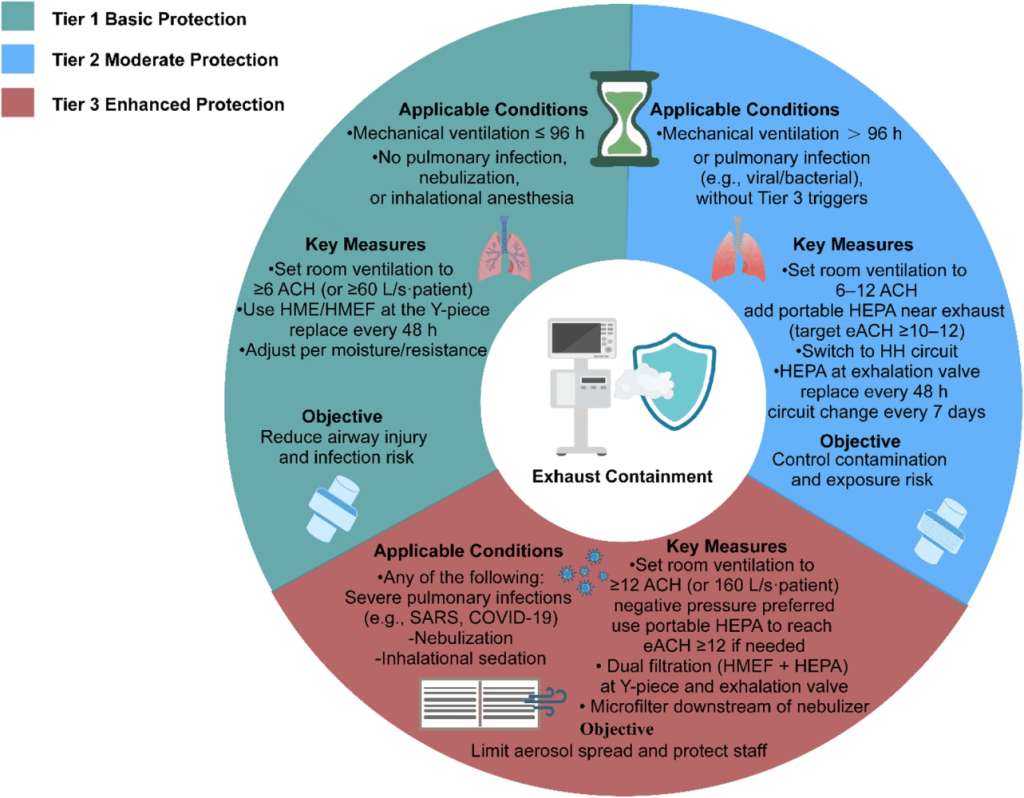
9️⃣ “Three-tier prevention strategy” — a practical clinical roadmap
Shi et al. propose a graded approach (Fig. 5, page 15):
Tier 1 – Basic Protection
IMV ≤96 hours, no infection, no nebulization
- HME/HMEF at Y-piece
- standard circuit management
- ≥6 ACH room ventilation
Tier 2 – Moderate Risk
IMV >96 hours, or pulmonary infection
- switch to heated humidification
- add HEPA at exhalation valve
- circuit change every 7 days
- ≥10–12 effective ACH
Tier 3 – High-Risk Situations
SARS, COVID-19, nebulization + infection, inhaled anesthetics
- ≥12 ACH or negative pressure
- dual filtration (HMEF + HEPA)
- microfilter downstream of nebulizer
- strict closed-circuit handling
This is the first cohesive, risk-stratified framework specific to ventilator exhaust.
🔟 Future directions: digital monitoring, smarter filters, CFD-guided ICU design
The authors highlight innovations including:
- silver-impregnated antimicrobial filters
- graphene–silver biocidal nanocomposites
- eNose VOC analytics + AI for real-time exhaust monitoring
- CFD-based design of ICU airflow patterns
This represents the next frontier in environmental infection control.
4. How This Should Influence Your Practice
- Treat ventilator exhaust as a potential infectious stream, not a neutral outlet.
- Use HMEF or HEPA filtration consistently—especially during high-risk procedures.
- Eliminate circuit breaks whenever possible; use closed suctioning.
- Filter or contain nebulizer emissions.
- Advocate for robust room ventilation and portable HEPA augmentation.
- Recognize inhaled anesthetic waste as an occupational hazard.
- Consider adopting tiered protection protocols in daily workflow.
5. Bottom Line for Clinicians
We cannot see ventilator exhaust — but it sees us. Managing it is now part of modern critical care and infection prevention.
This review signals a paradigm shift: Ventilator exhaust management is feasible, evidence-supported, and essential for ICU safety.
6. Discussion Question
How should hospitals balance the need for exhaust filtration and direction with real-world constraints such as resistance limits, cost, and infrastructure?
We look forward to your insights.

Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.