Abstract
This study evaluated whether a newly developed smart nasal cannula can accurately and continuously measure tidal volume (VT) and minute ventilation (VE) in adults across a wide range of respiratory patterns using a validated benchtop model. The device uses nasal pressure and flow dynamics, derived from RPD (respiratory pressure difference) sensors, to estimate VT and VE without requiring a mask, spirometer, or ventilator connection.
Across high-fidelity simulated breathing profiles—including normal, obstructive, restrictive, rapid shallow breathing, sighing, and apnea—the cannula provided VT and VE measurements with clinically acceptable accuracy when compared to a gold-standard reference pneumotachograph. Although systematic bias increased at extremes of tidal volume and flow, the system showed strong correlation and stability across most conditions. The authors conclude that a non-invasive, wearable cannula-based monitoring tool may allow real-time ventilation monitoring outside the ICU, including on wards, in transport, and in resource-limited settings.
Key Insights
1️⃣ Concept: A nasal cannula that functions like a ventilator flow sensor
The smart cannula integrates pressure sensors capable of detecting airflow pattern changes at the nares. By processing RPD signals, it estimates VT and VE continuously without requiring a mask, spirometry interface, or tight seal—potentially transforming monitoring outside critical care environments.
2️⃣ Testing approach: High-fidelity bench simulation of real-world breathing
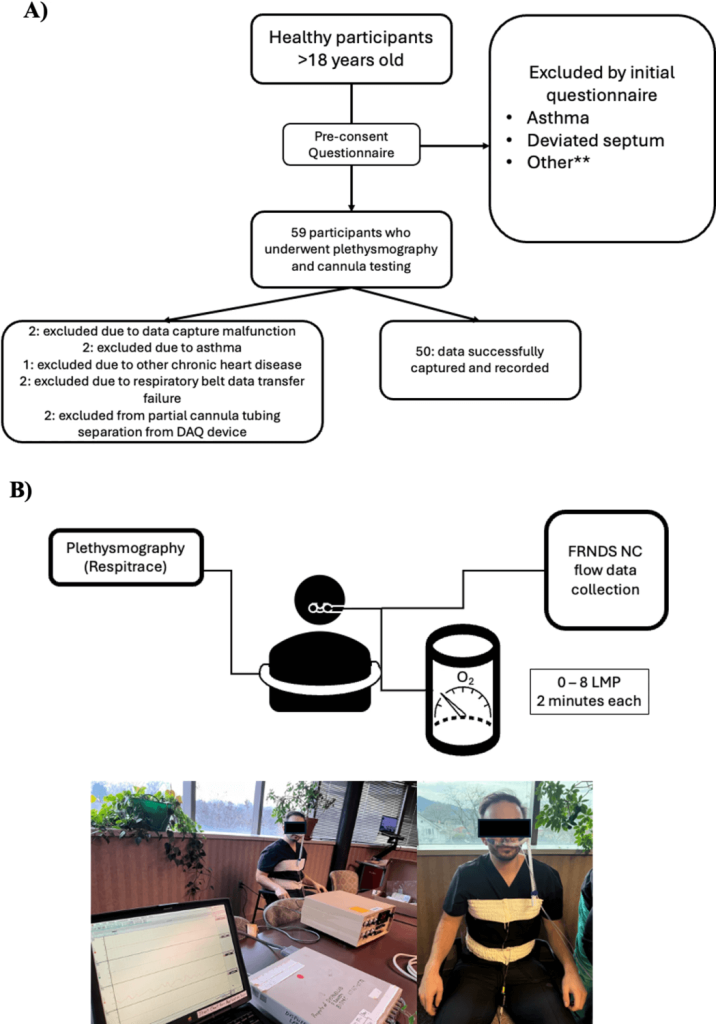
The study used a validated ASL 5000 test lung to simulate multiple adult respiratory patterns: normal breathing, obstructive lung disease, restrictive mechanics, tachypnea, bradypnea, rapid shallow breathing, sigh breaths, and apnea. Each was tested across multiple VT and RR ranges to assess accuracy across physiologic extremes.
3️⃣ Reference standard: Gold-standard pneumotachograph
All measurements from the smart cannula were compared to a high-precision reference pneumotachograph, allowing direct assessment of error, bias, and correlation under controlled conditions.
4️⃣ Accuracy: Strong correlation with reference measurements
Across the majority of simulated conditions, the smart cannula showed excellent correlation between measured and true VT/VE. Mean bias was small during normal and moderately abnormal breathing, indicating reliable measurement fidelity during typical clinical scenarios.
5️⃣ Performance limitations appear at extremes
Accuracy decreased when tidal volumes were extremely small (<200 mL), extremely large (>800 mL), or when airflow patterns produced very high turbulence (e.g., severe obstructive profiles). This reflects the known challenge of nasal-based systems in capturing extremes of flow.
6️⃣ Minute ventilation remained highly reliable across conditions
Even when VT accuracy diminished under extreme conditions, VE (which integrates rate × VT) maintained clinically useful precision. This suggests the device could be valuable for early detection of hypoventilation or hyperventilation on the wards.
7️⃣ Device stability is promising for continuous monitoring
Signal drift was minimal, and the RPD sensors demonstrated consistent output across long testing periods. This is critical for real-time monitoring applications, where drift can compromise decision-making.
8️⃣ Major clinical use-case: Early detection of deterioration outside the ICU
Because the device is non-invasive, wearable, and does not require patient cooperation, it may enable early respiratory deterioration detection in:
- step-down units
- EDs
- peri-procedural sedation
- hospital ward patients at risk for opioid respiratory depression
- interhospital transport
These are areas where ventilatory monitoring is currently sparse or absent.
9️⃣ Potential value in high-flow and NIV settings
Although not tested in vivo, the system architecture is compatible with environments where mask interfaces disrupt traditional minute ventilation monitoring. This opens the door for future validation during high-flow nasal cannula therapy, which currently lacks reliable VT/VE monitoring tools.
🔟 Study limitation: Benchtop only—clinical trials are needed
The authors emphasize that real-world patient studies are required to validate performance under conditions involving variable nasal anatomy, leaks, secretions, patient movement, and temperature/humidity changes.
🧩 Clinical Takeaways
- This smart cannula could address one of the biggest gaps in modern monitoring: continuous, non-invasive ventilation assessment outside the ICU.
- VT and VE accuracy is strong across normal physiologic ranges.
- VE appears more robust than VT under extreme conditions.
- Early deterioration detection may become possible without masks, spirometers, or ventilator circuits.
- Real-world clinical validation remains essential before widespread adoption.
Conclusion
This study demonstrates that a smart nasal cannula can accurately and continuously estimate tidal volume and minute ventilation across a wide range of simulated breathing patterns. While benchtop-only, the findings highlight a promising direction in respiratory monitoring—one that could extend accurate ventilation assessment into settings where traditional tools are impractical.
If validated clinically, this technology may fundamentally broaden our capacity to detect respiratory failure early, monitor ventilation non-invasively, and improve patient safety beyond the ICU.
Discussion Question
Could continuous, cannula-based ventilation monitoring become a new vital sign in hospitals—equivalent in importance to pulse oximetry?

Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.

