Abstract:
The approach to renal replacement therapy (RRT) in critically ill patients has evolved from early, empirical use to more individualized, evidence-based practice. Large randomized controlled trials (RCTs) now support delayed initiation unless urgent complications arise, and new data inform decisions on modality, dose, and discontinuation. This review summarizes the key updates and current standards of care for RRT in the ICU.
Key Insights:
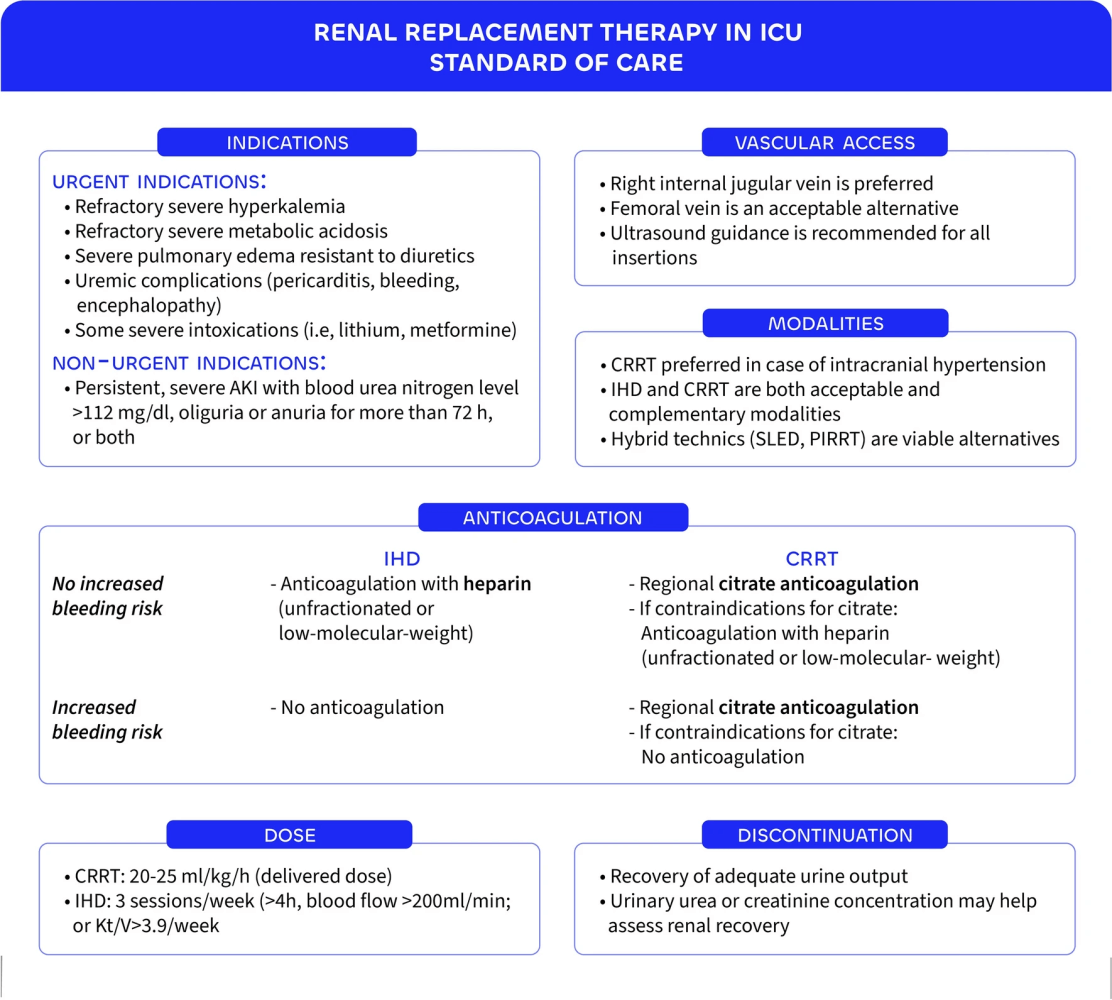
- RRT should be initiated only for urgent indications: severe hyperkalemia, refractory metabolic acidosis, fluid overload causing pulmonary edema, or uremic complications such as encephalopathy or pericarditis .
- In the absence of these emergencies, a delayed initiation strategy with close monitoring is safe, avoiding unnecessary RRT in up to 40% of patients .
- Excessive delays beyond 3 days of oligo-anuria or serum urea >40 mmol/L increase mortality, emphasizing balanced timing .
- CRRT is generally preferred in hemodynamically unstable patients, but evidence does not consistently show survival benefits over intermittent hemodialysis (IHD) .
- Observational data suggest CRRT may reduce dialysis dependence at 90 days, though some analyses even link it to higher mortality, highlighting ongoing uncertainty .
- Hybrid techniques such as SLED or PIRRT and peritoneal dialysis remain viable, especially in resource-limited settings .
- Dose intensity matters: higher RRT doses show no survival benefit and may delay renal recovery, while very high-volume hemofiltration offers no clinical advantage .
- Current recommendations target a CRRT dose of 20–25 mL/kg/h, while IHD or PIRRT should aim for a weekly Kt/V of approximately 3.9 .
- Discontinuation should be attempted once urine output exceeds 500 mL/h spontaneously or 2.4 L/day with diuretics, balancing risk of premature withdrawal against harm from prolonged RRT .
- Future practice will move toward personalized RRT guided by biomarkers (NGAL, proenkephalin, CCL14) and predictive modeling to optimize initiation, modality, and discontinuation .
Conclusion:
The standard of care for RRT in the ICU now emphasizes indication-driven initiation, individualized modality choice, cautious dosing, and timely discontinuation. Personalized approaches informed by biomarkers and modeling tools are likely to shape the next generation of RRT strategies.
Take-Home for Clinicians: Avoid reflexive early RRT initiation unless urgent complications are present. Tailor modality and dosing to the patient’s hemodynamic profile and resource context. Prioritize urine output and clinical trajectory when considering discontinuation.
Discussion Question: Will the integration of biomarkers and personalized predictive models finally resolve the debate on RRT initiation timing and modality in the ICU?

Take Advantage of This Resource
I encourage you to explore this growing library of articles and leverage it to stay informed on the latest in critical care. Visit the collection today at: https://perfusfind.com/ic/
This is another step in making high-quality, evidence-based information easily accessible to the critical care community. As always, thank you for your continued support!
As always, don’t forget to like, share, and subscribe. See you on the other side!
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.