Organ Crosstalk in Sepsis-Related ALI/ARDS: New Insights into Lung–System Interactions
Abstract: Sepsis-related ALI/ARDS remains highly lethal (>30% mortality), and most research has historically focused only on the lung. This review highlights organ crosstalk as a central mechanism in sepsis pathophysiology. The authors differentiate pulmonary sepsis-associated ALI/ARDS (PSA) from extrapulmonary sepsis-associated ALI/ARDS (ESA), outline systemic inflammation, microbiome dynamics, and cell-death cascades, and emphasize extracellular vesicles (EVs) as key mediators and potential therapeutic targets.
Key Insights
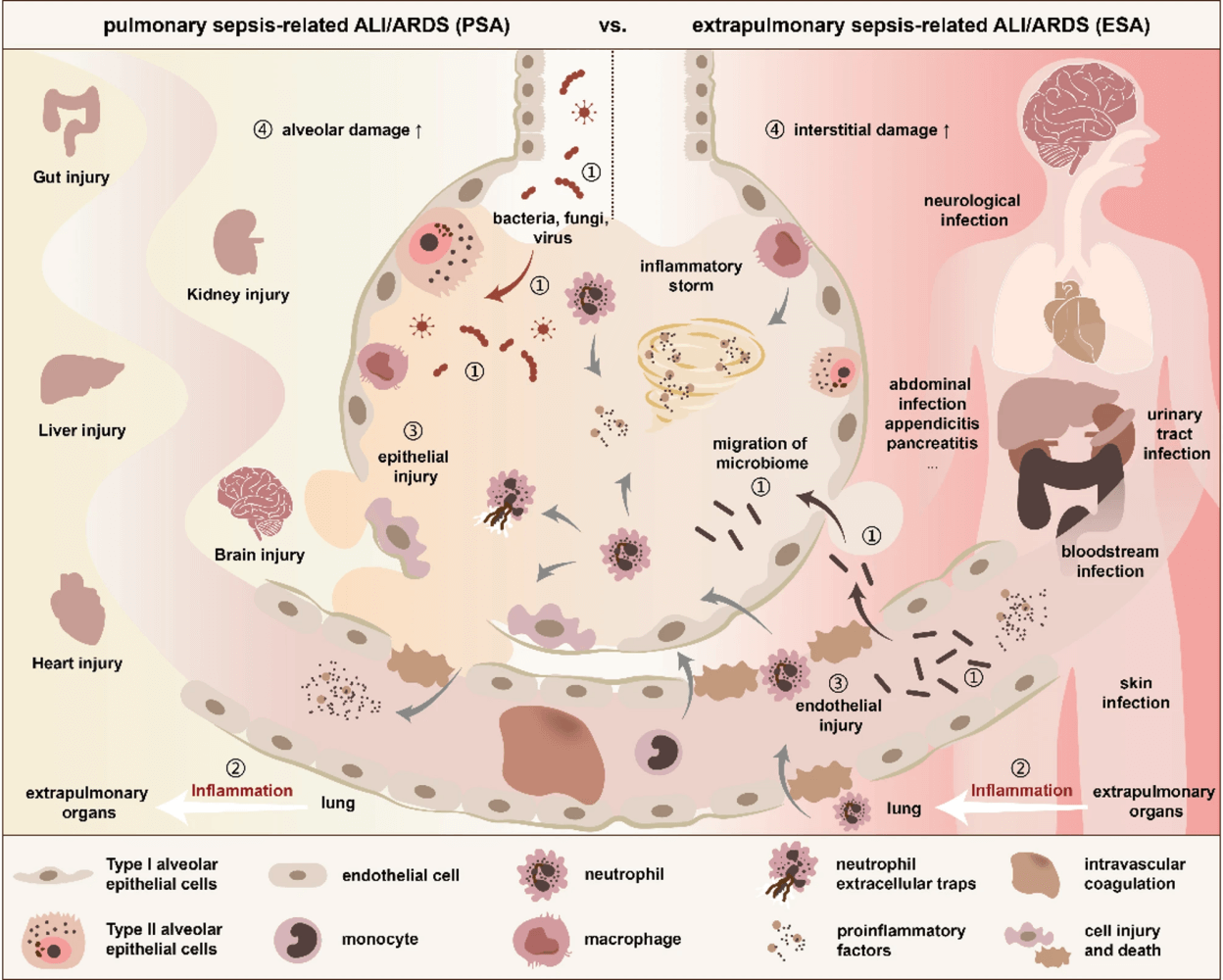
- PSA vs ESA distinctions: PSA arises from lung infections, leading to epithelial injury and consolidation, while ESA results from extrapulmonary infections, causing endothelial injury, interstitial changes, and ground-glass opacities.
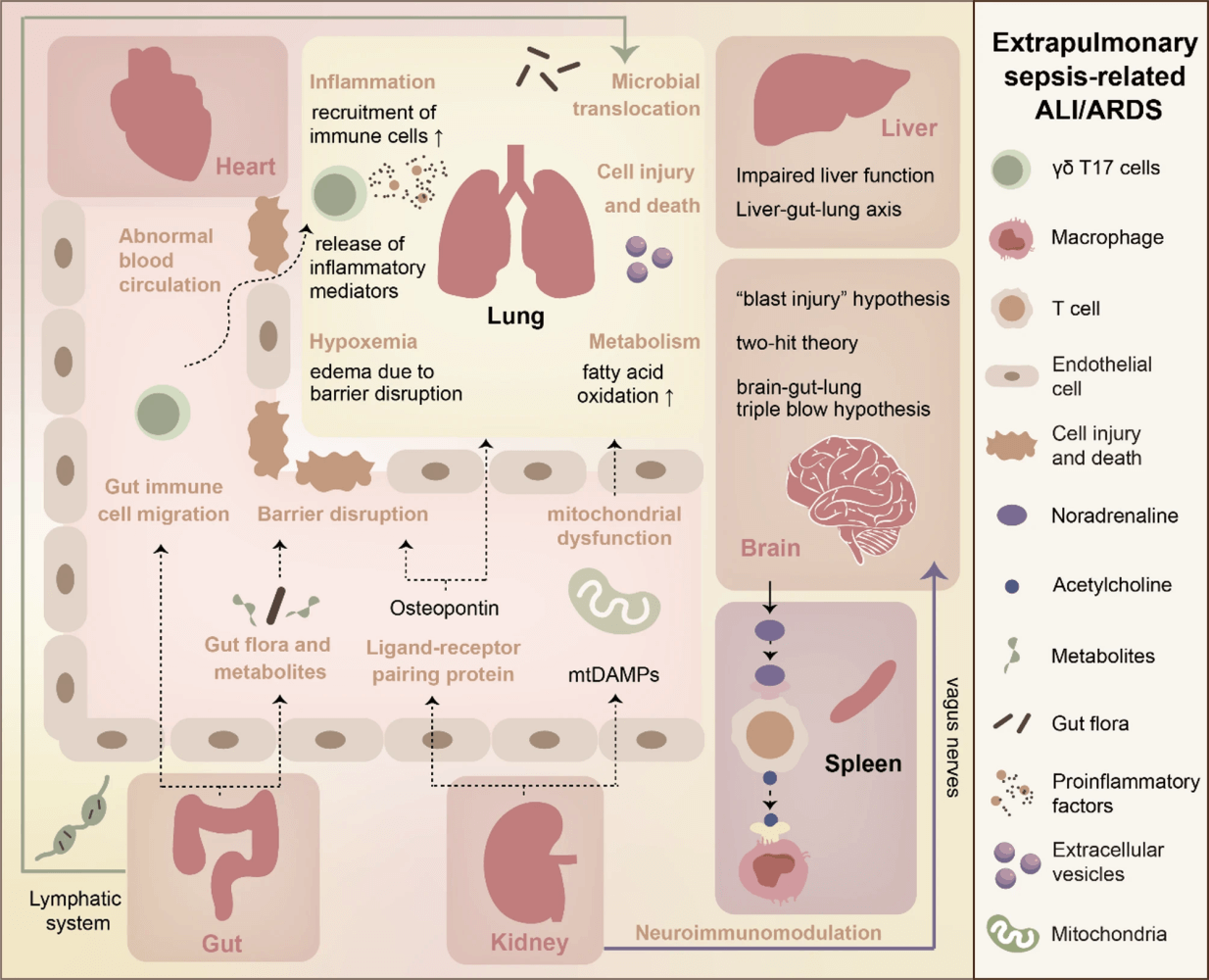
- Systemic inflammation: Shared mechanism involves cytokines (TNF-α, IL-6), endothelial activation, coagulation, microthrombosis, and hypoperfusion; but organ-specific responses (liver detoxification loss, neuroendocrine signaling, splenic cytokine release) amplify crosstalk.
- Microbiome roles: Dysbiosis and barrier failure enable gut–lung microbial translocation, while lung microbiota can affect the brain and distant organs. Metabolites (e.g., butyrate, LPS) regulate immunity and barrier integrity.
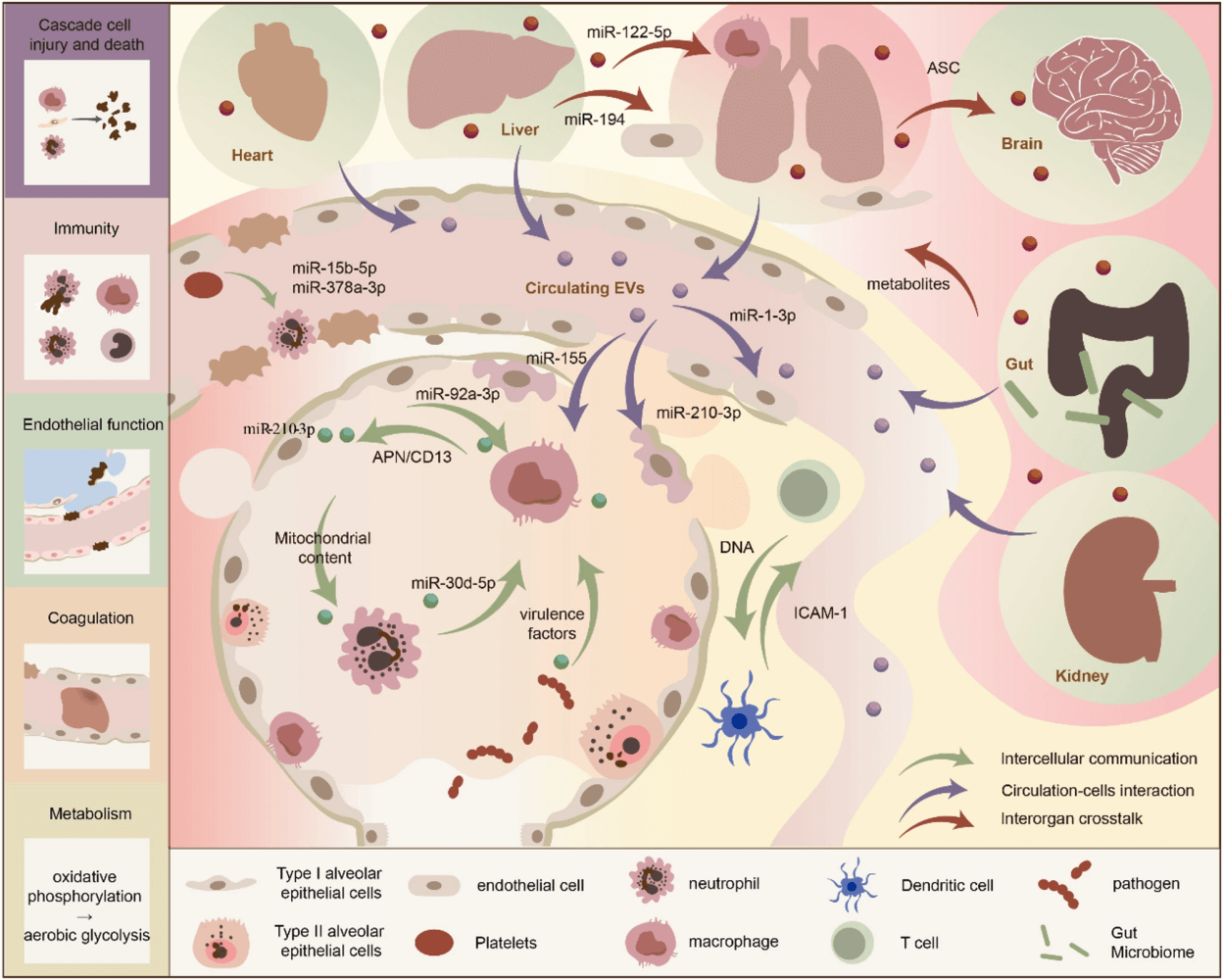
- Cascade of cell injury: Cell death modes (necrosis, necroptosis, apoptosis, pyroptosis, NETosis) propagate damage across organs via EVs, ROS, and DAMPs. Domino-like injury cascades may explain multi-organ persistence post-sepsis.
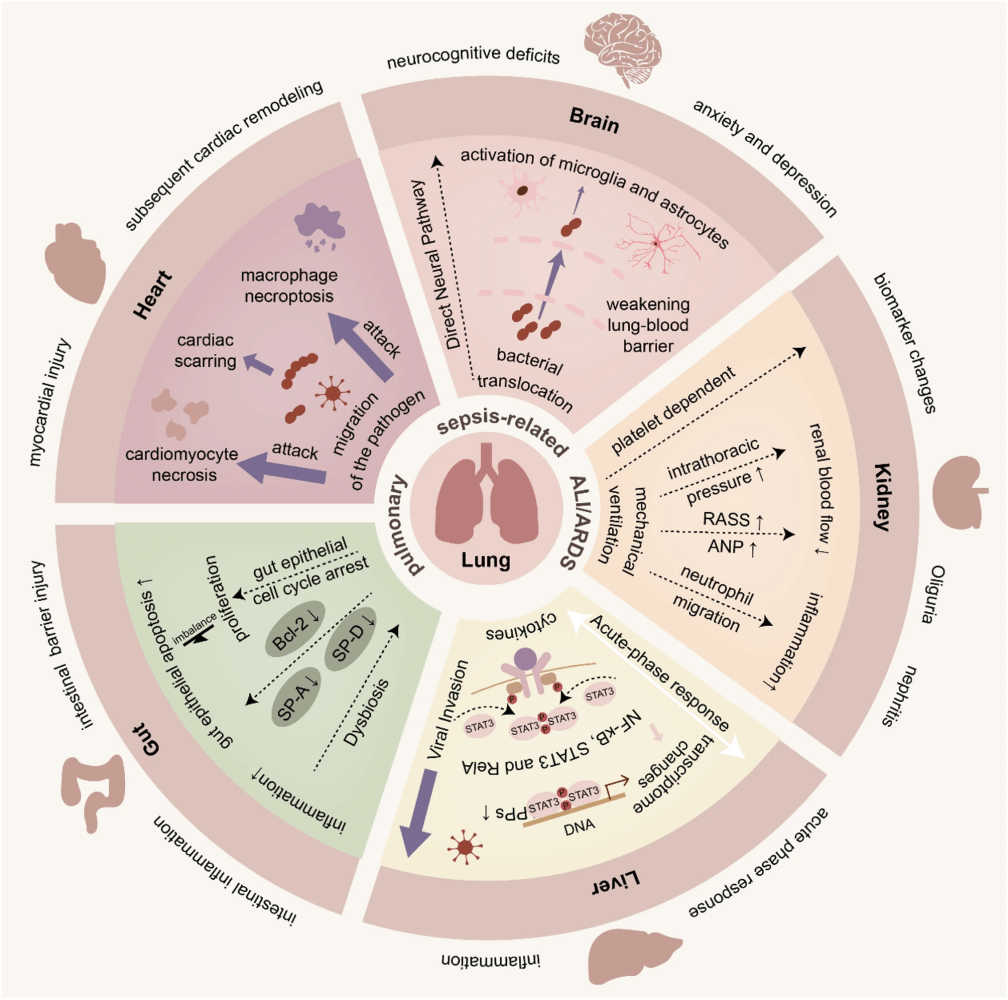
- Lung–brain axis: BBB disruption, microbial translocation, microglia activation, and TRPV1+ sensory neurons mediate neurological complications post-ARDS, including depression and cognitive decline.
- Lung–kidney axis: Pneumonia can induce AKI via platelet-mediated injury; mechanical ventilation increases AKI risk through hemodynamics and inflammation. Conversely, AKI triggers osteopontin-CD44 signaling, damaging lung endothelium.
- Lung–gut axis: Pneumonia induces gut epithelial apoptosis and barrier dysfunction, which in turn worsens pulmonary inflammation through bacterial translocation and reduced Tregs/SCFAs.
- Lung–liver axis: Viral invasion, Kupffer cell dysfunction, and acute-phase protein release enhance systemic inflammation; dysregulated hepatopulmonary signaling aggravates ALI/ARDS.
- Lung–heart axis: Pathogens can directly invade myocardium; inflammation and mitochondrial dysfunction drive cardiopulmonary injury.
- Extracellular vesicles (EVs): EVs mediate immunity, barrier dysfunction, metabolism, and coagulation in crosstalk. Higher circulating EVs correlate with sepsis severity and mortality, highlighting their promise as biomarkers and therapeutic targets.
Why This Matters
Viewing sepsis-related ALI/ARDS through the lens of multi-organ communication shifts the paradigm from isolated lung injury to systemic interplay. This could open doors for multi-targeted therapies—not just ventilatory or anti-inflammatory interventions, but microbiome modulation, EV-targeting, and organ-protective strategies.
Conclusion
Crosstalk between the lungs and other organs is central to sepsis-related ALI/ARDS. While challenges remain—heterogeneity, limited animal models, and trial feasibility—emerging omics, organ-on-chip systems, and 3D organoids promise better mechanistic insights and future therapeutic breakthroughs.
Take-Home for Clinicians
- Recognize sepsis-related ALI/ARDS as a multi-organ syndrome.
- Pay attention to organ-specific interactions (lung–gut, lung–kidney, lung–brain).
- Watch for EV-based biomarkers and microbiome-targeted therapies in coming years.
Discussion Question: Will targeting organ crosstalk mechanisms (EVs, microbiota, neuroendocrine signaling) become the next frontier in sepsis-related ALI/ARDS therapy?

Take Advantage of This Resource
I encourage you to explore this growing library of articles and leverage it to stay informed on the latest in critical care. Visit the collection today at: https://perfusfind.com/ic/
This is another step in making high-quality, evidence-based information easily accessible to the critical care community. As always, thank you for your continued support!
As always, don’t forget to like, share, and subscribe. See you on the other side!
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.

