Summary
This comprehensive review explores why emergency airway management remains one of the most dangerous procedures in critical care medicine. Despite decades of improvement in first-pass success, the physiologic burden of induction and immediate transition to positive-pressure ventilation continues to precipitate hypotension, shock, and cardiac arrest in a large proportion of critically ill patients. The authors dissect the epidemiology, mechanisms, modifiable risks, and evidence-based strategies that clinicians can use to mitigate cardiovascular collapse in the peri-intubation period.
Key Insights
1. Peri-intubation hypotension is common and deadly.
The review reports that ≥40% of critically ill patients experience hypotension during or immediately after intubation, and these episodes strongly correlate with multi-organ failure, cardiac arrest, and mortality, even when brief or modest in degree.
2. Multiple risk factors converge to amplify vulnerability.
Shock, hypoxemia, right ventricular dysfunction, severe acidosis, metabolic collapse, pregnancy, and obesity all heighten physiologic fragility, while anatomic airway difficulty increases the likelihood of multiple attempts and cumulative hemodynamic stress.
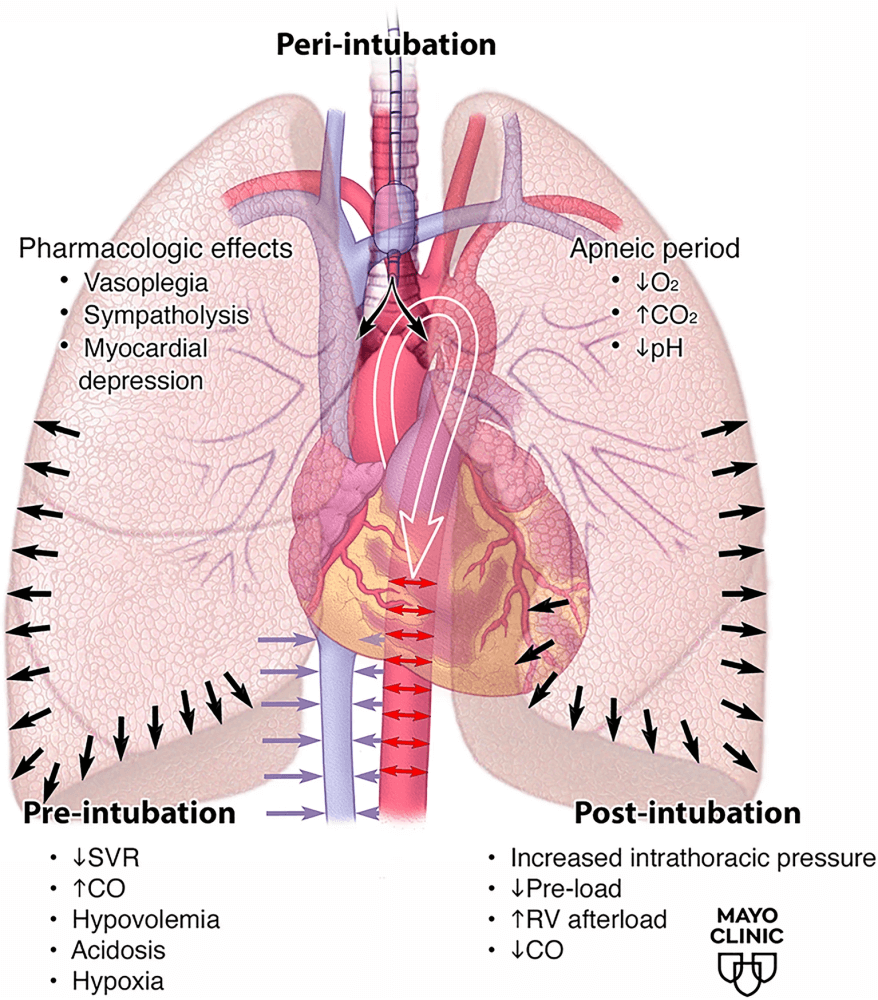
3. The transition from negative to positive-pressure ventilation is a major driver of collapse.
Loss of spontaneous breathing eliminates negative intrathoracic pressure, reducing venous return and preload. Meanwhile, positive-pressure ventilation increases RV afterload and may precipitate acute RV failure—especially in pulmonary hypertension or shock. The diagram on page 5clearly illustrates the simultaneous effects of vasoplegia, apnea, rising CO₂, falling pH, and loss of cardiac output.
4. Risk stratification tools exist but are underused.
HYPS ≥2, shock index ≥0.7–0.8, or modified shock index ≥0.9 strongly predict post-intubation hypotension and should trigger anticipatory hemodynamic optimization. Yet, many clinicians still do not systematically screen for physiologic difficulty.
5. Preoxygenation must evolve beyond traditional methods.
Noninvasive ventilation (NIV) and high-flow nasal cannula outperform simple mask oxygenation. Randomized trials show bag-mask ventilation during the apneic period reduces hypoxemia without increasing aspiration risk. The review emphasizes that NIV may unmask hemodynamic instability “in a controlled fashion,” allowing correction before induction.
6. Routine fixed-volume fluid boluses are not supported by evidence.
Two major trials (PREPARE I & II) showed no reduction in cardiovascular collapse with 500-mL boluses. Fluid resuscitation should be individualized—beneficial for true hypovolemia but unnecessary or harmful in patients who are not fluid-responsive.
7. Push-dose vasopressors help—but evidence remains limited.
Phenylephrine, epinephrine, or ephedrine can provide rapid MAP augmentation, serving as a bridge until continuous norepinephrine infusions take effect. However, medication-error risk is significant without standardized preparation and pharmacist involvement.
8. Prophylactic vasopressor infusions are increasingly used—despite sparse evidence.
Early norepinephrine initiation may stabilize patients before induction, but high-quality evidence is still pending (PREVENTION & FLUVA trials). Peripheral vasopressor administration is considered safe when monitored.
9. Induction agent selection must be individualized—not dogmatic.
Etomidate remains hemodynamically neutral but may suppress adrenal function. Ketamine supports sympathetic tone unless the patient is catecholamine-depleted. Reduced-dose induction is widely used, but evidence is inconsistent, and risks include awareness under paralysis.
10. ECMO may be the safest option for the highest-risk airway cases.
In patients with profound shock, severe pulmonary hypertension, or expected loss of perfusion with induction, pre-intubation ECMO cannulation—or ECMO standby—may be lifesaving. The clinical algorithm on page 11provides a stepwise framework for identifying moderate- to high-risk patients and escalating interventions appropriately.
Conclusion
Peri-intubation cardiovascular collapse remains a major, preventable cause of morbidity and mortality in critical care. This review makes one message unequivocally clear: safe airway management goes far beyond technical skill. Clinicians must recognize physiologic difficulty, resuscitate before inducing, anticipate hemodynamic decline, individualize medication choice, and escalate to advanced support—including ECMO—when necessary. A structured, physiology-driven, bundle-based approach is essential for improving outcomes.
Discussion Questions
- Should vasopressor infusions become the standard of care before induction in any patient with SI ≥ 0.7, or does this risk overtreatment?
- How can airway teams incorporate the HYPS or shock index into real-time workflows without adding cognitive burden during emergencies?
- Would bundled peri-intubation protocols (NIV preoxygenation, early pressors, arterial line placement, reduced-dose induction) reduce cardiovascular collapse more effectively than isolated interventions?

Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.

