ARDS: Pathophysiological Insights, Subphenotypes, and Precision Medicine
Abstract: ARDS is a syndrome of hypoxemic respiratory failure caused by alveolo-capillary barrier dysfunction, dysregulated inflammation, and mechanical injury. Despite 60 years of research, mortality remains near 40%. The heterogeneity of ARDS has driven interest in subphenotypes—biological, clinical, physiological, and radiographic—that may explain variable responses to therapies. This review summarizes current pathophysiological insights, describes key subphenotypes, and highlights implications for personalized therapeutic strategies.

Key Insights:
- Barrier dysfunction occurs when epithelial and endothelial injury increase permeability, leading to protein-rich edema, impaired clearance, and neutrophil/platelet-driven inflammation.
- Inflammation is driven by neutrophils, macrophages, and cytokines such as IL-1β, IL-6, IL-8, and TNF-α, which propagate both local damage and systemic MODS, underscoring that ARDS is more than a lung disease.
- Mechanical forces from ventilation cause volutrauma, barotrauma, atelectrauma, and biotrauma, amplifying injury. Driving pressure is the strongest ventilatory predictor of mortality.
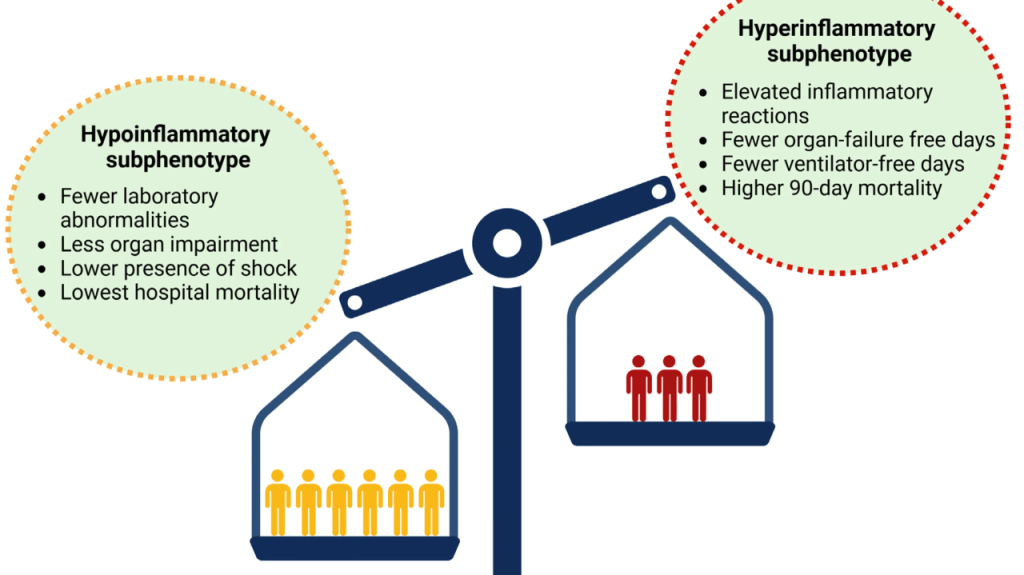
- Biological subphenotypes include the hyperinflammatory group, marked by high cytokines and vasopressor use with worse outcomes, and the hypoinflammatory group, with lower inflammation and better survival; they show distinct responses to fluids, PEEP, and statins.
- Clinical subphenotypes demonstrate that trauma-related and non-sepsis ARDS patients have better outcomes compared with sepsis-driven ARDS, while late-onset ARDS is linked to higher mortality.
- Physiological subphenotypes are defined by PaO₂/FiO₂ ratio, compliance, dead-space fraction, and recruitability, with low compliance and high dead-space fraction predicting mortality.
- Radiographic subphenotypes distinguish focal ARDS, characterized by dorsal opacities and strong responsiveness to prone positioning, from non-focal ARDS, which presents diffuse opacities and is recruitable with PEEP. Misclassification, as highlighted in the LIVE trial, undermines precision benefits.
- Cardiovascular subphenotypes identified by echocardiography reveal distinct patterns of right ventricular dysfunction, with RV impairment associated with mortality exceeding 70%.
- Therapeutics vary by subphenotype; low tidal volumes and optimized PEEP remain standard, but fluid restriction is beneficial only in hyperinflammatory patients, and simvastatin improved survival in hyperinflammatory ARDS in HARP-2 while rosuvastatin failed in SAILS.
- Future directions include biomarker-driven trials, mesenchymal stromal cell therapy, and biologics such as anti-IL-6, anti-TNF, and IRAK4 inhibitors, though bedside phenotyping remains the barrier to precision medicine.
Conclusion: ARDS is not a single disease but a heterogeneous syndrome. Subphenotypes influence outcomes and therapeutic responses, and their accurate identification may finally unlock precision therapies. Standardized definitions, bedside biomarkers, and phenotype-specific trials are essential for progress.

Take-Home for Clinicians: ARDS should always be approached as a heterogeneous entity, with careful attention to phenotype-specific responses to fluids, PEEP, and adjunct therapies. Trials that incorporate biological and physiological classifications are needed to bring precision medicine into daily practice.
Discussion Question: How can rapid subphenotype identification be brought to the bedside so ARDS therapies can be truly individualized?

Take Advantage of This Resource
I encourage you to explore this growing library of articles and leverage it to stay informed on the latest in critical care. Visit the collection today at: https://perfusfind.com/ic/
This is another step in making high-quality, evidence-based information easily accessible to the critical care community. As always, thank you for your continued support!
As always, don’t forget to like, share, and subscribe. See you on the other side!
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.

