Ultrafiltration During Cardiopulmonary Bypass: Enhanced Review of Clinical Evidence and Application
- Home
- Cannulation
- Current Page
1. Introduction
Cardiopulmonary bypass (CPB) leads to pronounced hemodilution and provokes a systemic inflammatory response from blood interaction with artificial surfaces (Ziyaeifard et al., 2014) (OAText, Lippincott Journals). Ultrafiltration (UF) mitigates these effects by hemoconcentrating the blood—removing plasma water and low molecular‑weight substances, while conserving red blood cells, platelets, proteins, and coagulation factors (ScienceDirect Topics) (ScienceDirect, JTCVS).
2. Types of Ultrafiltration
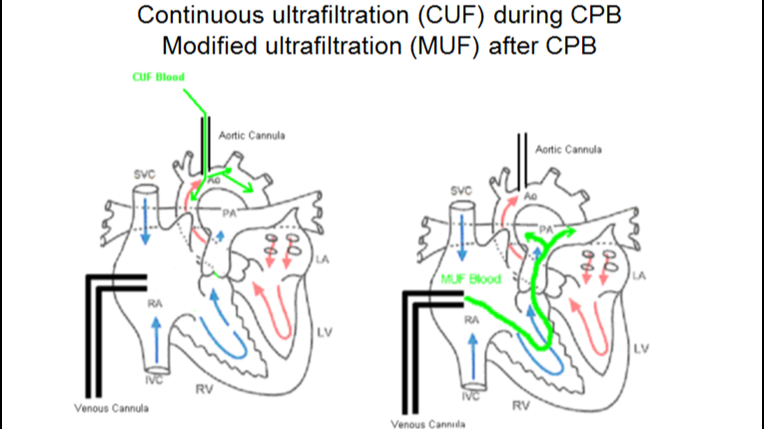
2.1 Conventional Ultrafiltration (CUF)
Performed during CPB, CUF continuously filters venous reservoir blood through a hemoconcentrator, thus limiting fluid overload and slightly elevating hematocrit. CUF is often combined with MUF in pediatric surgery to enhance efficacy (Hu et al., 2021) (Lippincott Journals).
2.2 Modified Ultrafiltration (MUF)
Applied immediately after CPB discontinuation, MUF draws arterial blood via the aortic cannula and returns it through the venous side. In pediatric patients, MUF has been shown to substantially increase hematocrit and mean arterial pressure, while reducing inflammatory mediators (Ziyaeifard et al., 2014; modified ultrafiltration review) (PubMed). In adults, MUF has been associated with lower perioperative morbidity and reduced transfusion requirements (Luciani et al., 2001; modified ultrafiltration reduces morbidity) (American Heart Association Journals, OAText). However, while it lowers cytokine and adhesion molecule levels, corresponding improvements in clinical endpoints are inconclusive (Modified ultrafiltration beneficial for adults) (JCVA Online).
2.3 Continuous Intra‑operative Ultrafiltration
Continuous UF techniques (e.g. zero‑balance ultrafiltration) run throughout CPB aiming to maintain fluid and inflammatory balance. A recent systematic review and meta‑analysis involving 989 adult patients found that continuous UF did not reduce operative mortality but significantly reduced ICU length of stay by 7 hours, ventilation time by ~2 hours, and pneumonia incidence by 67% (Bierer et al., 2023) (PubMed). Earlier trials also noted reduced postoperative bleeding and transfusion needs, along with shortened hospitalization (prospective study) (ScienceDirect).
3. Evidence in Pediatric and Adult Populations
3.1 Pediatric Evidence
A meta‑analysis of eight randomized controlled trials (405 pediatric patients) comparing CUF+MUF vs CUF alone revealed that combination UF significantly increased post‑CPB hematocrit (SMD = 1.85; 95% CI 0.91–2.79) and total ultrafiltrate volume (SMD = 1.46; 95% CI 0.51–2.41) (Hu et al., 2021) (Lippincott Journals). However, differences in clinical outcomes such as ventilation duration, ICU stay, and blood loss were unclear due to heterogeneous sensitivity analyses (Hu et al., 2021) (Lippincott Journals). Notably, high‐heterogeneity (I² 88–90%) across studies limits firm conclusions on hemodynamics, chest drainage, and length of stay (Hu et al., 2021) (Lippincott Journals).
3.2 Adult Evidence
A broader adult meta‑analysis (22 studies, 8,538 patients) demonstrated that UF reduced perioperative bleeding (~108 mL), red blood cell transfusion (~0.76 units/patient), and ICU stay (~0.16 days), though it did not affect major cardiovascular outcomes or mortality (systematic review) (OAText). A randomized trial also reported reduced early morbidity and lower blood product use with MUF (Luciani et al., 2001) (American Heart Association Journals). Meanwhile, continuous UF strategies showed recovery benefits, as described above (Bierer et al., 2023) (PubMed).
4. Mechanisms Underlying Benefits
- Hemoconcentration: UF elevates hematocrit and plasma protein concentration, improving oxygen delivery and reducing tissue edema (Ziyaeifard et al., 2014) (Lippincott Journals).
- Inflammation modulation: Removes low‑molecular weight inflammatory mediators such as IL‑6, IL‑8, TNF‑α, and adhesion molecules (modified ultrafiltration overview) (ResearchGate).
- Blood conservation: Decreases the need for blood transfusions and reduces postoperative chest drainage (systematic adult review; pediatric meta‑analysis) (OAText).
5. Limitations, Risks, and Quality of Evidence
- Operative time: MUF may slightly prolong total CPB time and add complexity to perfusion management (Hu et al., 2021) (Lippincott Journals).
- Potential complications: Hypotension, hemolysis, air embolism, and cannula obstruction are recorded risks—especially with aggressive UF in small infants (discussions in pediatric studies) (Lippincott Journals).
- Renal concerns: Excessive UF volume may impair renal perfusion, though adult meta‑analyses did not reveal significant differences in acute kidney injury (adult review) (OAText, PubMed).
- Evidence quality: Most studies show moderate to high heterogeneity; randomized evidence in adults remains limited and endpoints vary across trials, lowering overall certainty (Bierer et al., 2023) (BioMed Central).
6. Recommendations and Clinical Integration
- In paediatric cardiac surgery, the combined use of CUF and MUF is well-supported, demonstrating clear gains in hemoconcentration and modulating fluid balance.
- In adult cases, standard UF (CUF/MUF) modestly reduces bleeding and transfusion needs; continuous UF offers further advantages in reducing ICU stay, ventilation time, and pneumonia, albeit with low to very low quality of evidence.
- Practice considerations: UF implementation should account for patient age, procedure complexity, priming volume, co-morbidities, institutional protocols, and available perfusionist expertise.
Asif Mushtaq: Chief Perfusionist at Punjab Institute of Cardiology, Lahore, with 27 years of experience. Passionate about ECMO, perfusion education, and advancing perfusion science internationally.
