Substrate Enhancement in Cardioplegia: Current Concepts and Future Directions
- Home
- Cannulation
- Current Page
Introduction
Myocardial protection during cardiac surgery is a critical determinant of patient outcomes. Traditional cardioplegia techniques primarily aim to arrest the heart, reduce metabolic demand, and preserve myocardial viability (Buckberg, 1979). However, emerging strategies focus on augmenting the metabolic substrate supply during ischemia, an approach termed «substrate enhancement» (Fazel et al., 2004). By optimizing myocardial metabolism during ischemia-reperfusion periods, substrate-enhanced cardioplegia aims to better preserve myocardial energy stores, minimize reperfusion injury, and improve postoperative recovery (Hearse, 1980)
Normal Myocardial Metabolism
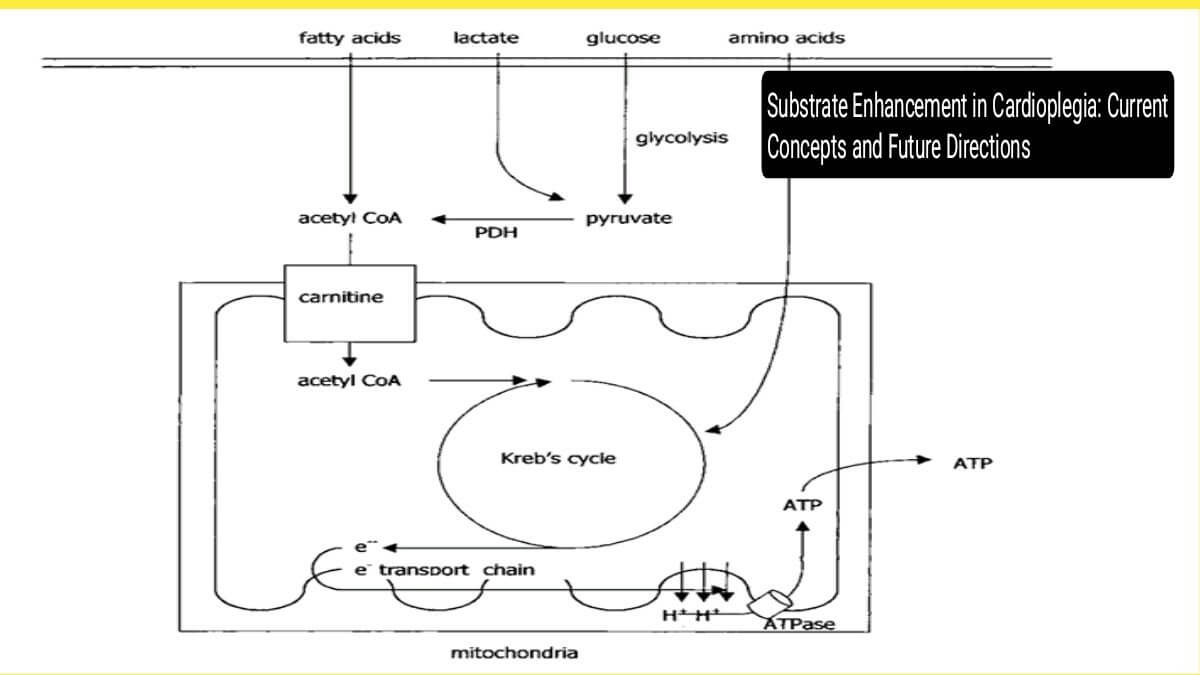
The myocardium under aerobic conditions derives approximately 60-70% of its energy from fatty acid oxidation and 30-40% from glucose oxidation (Stanley et al., 2005). Glucose enters the cardiomyocytes through glucose transporter (GLUT) proteins, mainly GLUT1 and GLUT4. These facilitate the transport of glucose across the cell membrane into the cytosol, where it undergoes glycolysis to produce pyruvate. In the presence of oxygen, pyruvate is decarboxylated by pyruvate dehydrogenase (PDH) to form acetyl-CoA, which enters the Kreb’s cycle. Oxidative phosphorylation within mitochondria generates ATP, the key energy currency. Under anaerobic conditions, however, pyruvate is converted to lactate, resulting in decreased ATP production and intracellular acidosis (Taegtmeyer, 1994).
Key Kreb’s cycle intermediates such as α-ketoglutarate and oxaloacetate also allow for the entry of amino acids like glutamate and aspartate into the cycle, supporting energy production even under stress (Opie, 1998).
Impact of Ischemia on Myocardial Metabolism
During ischemia, the cessation of oxygen supply shifts myocardial metabolism toward anaerobic glycolysis. This leads to:
- Rapid depletion of ATP.
- Accumulation of lactate and hydrogen ions.
- Activation of injurious pathways, including calcium overload and oxidative stress upon reperfusion (Jennings and Reimer, 1981).
Prolonged ischemia, even under hypothermic conditions, risks irreversible myocardial injury, underscoring the need for enhanced strategies to sustain cellular metabolism.
Strategies for Substrate Enhancement in Cardioplegia
Several substrate-based approaches have been developed to support myocardial metabolism during cardioplegic arrest:
- Glucose-Insulin-Potassium (GIK) Therapy. Enhances glycolytic flux and maintains PDH activity (Sodi-Pallares et al., 1962).Reduces free fatty acid concentration, minimizing lipotoxicity (Opie, 1998).Particularly effective in warm heart surgery but requires careful glucose control to avoid hyperglycemia-induced injury (Lopaschuk et al., 2002).
- Glutamate and Aspartate Supplementation. Serve as anaplerotic substrates to replenish Kreb’s cycle intermediates (Jennings and Reimer, 1981).Improve post-ischemic myocardial recovery in animal models; human clinical results are variable (Braunwald and Kloner, 1985).Administration may support better restoration of mitochondrial function.
- l-Carnitine SupplementationFacilitates the transport of long-chain fatty acids into mitochondria for β-oxidation (Ferrari et al., 1990). Reduces ventricular arrhythmias and enhances post-reperfusion myocardial function in experimental models. Human trials remain limited but promising (Davini et al., 1992).
- Histidine-Tryptophan-Ketoglutarate (HTK) SolutionA specialized crystalloid cardioplegic solution containing histidine for buffering, tryptophan for membrane stabilization, and ketoglutarate for metabolic support (Bretschneider, 1975). Demonstrated superior myocardial preservation in some clinical settings compared to traditional blood cardioplegia (Glauber et al., 1999).
- Adenosine SupplementationAdenosine acts as a potent vasodilator and anti-arrhythmic agent during ischemia. Enhances myocardial protection by promoting ATP preservation and improving microvascular perfusion (Lasley and Mentzer, 1995). May reduce reperfusion injury by attenuating neutrophil activation and oxidative stress

Figure : Schematic representation of key metabolic pathways influenced by substrate enhancement during cardioplegia. Glycolysis, Krebs cycle intermediates, and fatty acid transport are augmented to maintain ATP production and minimize ischemic injury.
Benefits and Challenges
Benefits:
- Enhanced recovery of contractile function.
- Reduced postoperative need for inotropic support (Buckberg, 1979).
- Lower incidence of arrhythmias and myocardial infarction.
- Potential improvement in long-term cardiac remodeling.
Challenges:
- Variability in clinical results due to differences in protocols, patient populations, and timing of substrate delivery.
- Risk of hyperglycemia with GIK therapy (Lopaschuk et al., 2002).
- Limited large-scale randomized controlled trials confirming the superiority of substrate-enhanced cardioplegia.
- Complexity in balancing multiple substrates without causing metabolic imbalance.
Future Perspectives
Future directions include:
- Precision substrate enhancement tailored to patient-specific metabolic profiles.
- Combining substrate enhancement with pharmacologic agents that modulate mitochondrial function (Stanley et al., 2005).
- Further exploration of gene and cell therapies to upregulate endogenous metabolic pathways.
- Conducting large, multicenter trials to establish standardized protocols.
The concept of «metabolic preconditioning» — priming the heart metabolically before ischemia — may also integrate into future myocardial protection strategies (Hausenloy and Yellon, 2004).
Conclusion
Substrate enhancement in cardioplegia represents a promising frontier in myocardial protection. By supporting the metabolic resilience of the myocardium during ischemia-reperfusion, these strategies aim to minimize injury and improve outcomes after cardiac surgery. While experimental studies are largely positive, further clinical validation is essential to define their precise role in routine practice.
References
- Braunwald, E. and Kloner, R.A., 1985. The stunned myocardium: prolonged, postischemic ventricular dysfunction. Circulation, 72(6), pp.953-967.
- Bretschneider, H.J., 1975. Myocardial resistance and tolerance to ischemia: physiological and biochemical basis. Journal of Cardiovascular Surgery, 16(3), pp.241-260.
- Buckberg, G.D., 1979. Strategies and logic of cardioplegic delivery to prevent, avoid, and reverse ischemic and reperfusion damage. Journal of Thoracic and Cardiovascular Surgery, 77(6), pp.803-815.
- Davini, P., Bigalli, A., Lamanna, F. and Scarlata, S., 1992. L-carnitine and propionyl-L-carnitine in cardiovascular diseases. Clinical Trials Journal, 29(3), pp.205-218.
- Fazel, S., Pelletier, M.P. and Goldman, B.S., 2004. Substrate Enhancement in Cardioplegia. In: Salerno, T.A. and Ricci, M., eds., Myocardial Protection. Blackwell Publishing, pp.94-118.
- Ferrari, R., Curello, S., Ceconi, C., Cargnoni, A., Pasini, E. and Albertini, A., 1990. Carnitine and carnitine derivatives in cardiovascular disease. Cardiovascular Drugs and Therapy, 4(4), pp.767-776.
- Glauber, M., Tenconi, S.M., Di Prima, A.L. and Bevilacqua, S., 1999. Histidine-tryptophan-ketoglutarate solution in myocardial protection: clinical results and cardioprotective mechanisms. Journal of Cardiothoracic and Vascular Anesthesia, 13(2), pp.171-179.
- Hausenloy, D.J. and Yellon, D.M., 2004. New directions for protecting the heart against ischemia-reperfusion injury: targeting the Reperfusion Injury Salvage Kinase (RISK)-pathway. Cardiovascular Research, 61(3), pp.448-460.
- Hearse, D.J., 1980. Myocardial protection during ischemia and reperfusion. Molecular and Cellular Biochemistry, 32(1), pp.161-177.
- Jennings, R.B. and Reimer, K.A., 1981. Lethal myocardial ischemic injury. American Journal of Pathology, 102(2), pp.241-255.
- Lasley, R.D. and Mentzer, R.M., 1995. Adenosine improves recovery of postischemic myocardial function by enhancing glycolysis. Circulation Research, 76(4), pp.678-686.
- Lopaschuk, G.D., Ussher, J.R., Folmes, C.D., Jaswal, J.S. and Stanley, W.C., 2002. Myocardial fatty acid metabolism in health and disease. Physiological Reviews, 82(1), pp.447-481.
- Opie, L.H., 1998. The Heart: Physiology from Cell to Circulation. 3rd ed. Lippincott-Raven.
- Sodi-Pallares, D., Testelli, M.R., Fishleder, B.L., Bisteni, A., Medrano, G.A., Friedland, C. and De Micheli, A., 1962. Effects of an intravenous infusion of a potassium-glucose-insulin solution on the electrocardiographic signs of myocardial infarction. American Journal of Cardiology, 9(2), pp.166-181.
- Stanley, W.C., Recchia, F.A. and Lopaschuk, G.D., 2005. Myocardial substrate metabolism in the normal and failing heart. Physiological Reviews, 85(3), pp.1093-1129.
- Taegtmeyer, H., 1994. Energy metabolism of the heart: from basic concepts to clinical applications. Current Problems in Cardiology, 19(2), pp.59-113.
Asif Mushtaq: Chief Perfusionist at Punjab Institute of Cardiology, Lahore, with 27 years of experience. Passionate about ECMO, perfusion education, and advancing perfusion science internationally.
