Optimizing Filtration in Cardiopulmonary Bypass: A Comprehensive Overview
- Home
- Cannulation
- Current Page
Introduction Cardiopulmonary bypass (CPB) plays a pivotal role in modern cardiac surgery by temporarily assuming the functions of the heart and lungs during complex procedures. However, despite its life-saving capabilities, CPB also introduces significant risks, including the potential for microembolism, systemic inflammatory response syndrome (SIRS), and ischemia-reperfusion injury. These complications are largely due to the foreign surfaces of the bypass circuit and the inflammatory responses triggered by cellular interactions during bypass.
One of the most effective ways to mitigate these risks is through filtration at various critical points in the CPB circuit. Filtration serves to remove harmful microemboli, particulate matter, and inflammatory mediators, all of which contribute to postoperative complications. The key areas where filtration is employed include the arterial line, cardioplegia line, cardiotomy reservoir, and the pre-bypass circuit. Each of these plays a vital role in protecting the patient from adverse effects during CPB (Gravlee et al., 2015).
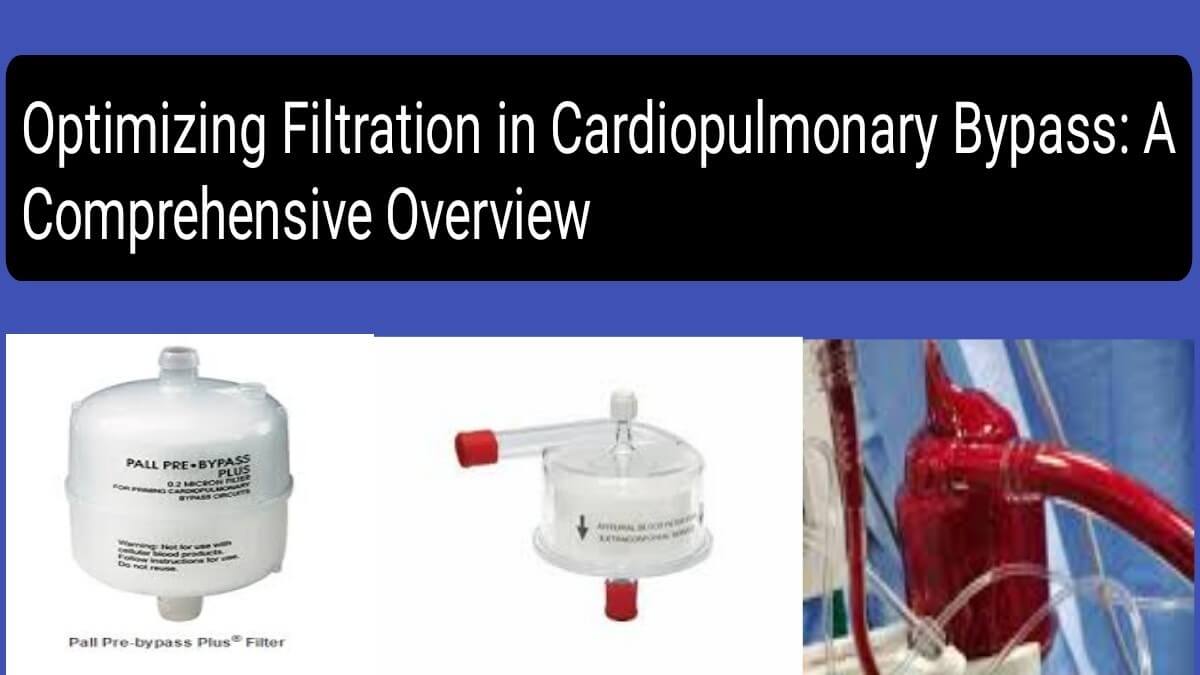
Pre-Bypass Filtration Pre-bypass filtration is one of the first lines of defense against contaminants in the prime solution, which is typically a balanced crystalloid or other solutions used to prime the CPB circuit. Pre-bypass filters are not used for filtering the blood prime. The blood prime contains necessary blood components, such as red blood cells, plasma, and platelets, and using a filter on it would eliminate essential cellular elements.
Instead, pre-bypass filters are employed specifically for filtering the crystalloid or other prime solutions before they enter the circuit. These filters are designed to capture particles, bacteria, and other contaminants that may have entered the circuit during assembly or transport. They are crucial to preventing contamination of the CPB system and avoiding adverse reactions from particulate matter entering the circuit. Pre-bypass filters typically remove impurities as small as 0.2 microns, such as bacteria and endotoxins, to reduce the risk of inflammatory reactions during bypass (Gravlee et al., 2015).
These filters, made from materials like microporous membranes, help ensure the purity of the prime solution. In doing so, they prevent any potential embolic events or inflammatory responses that might occur when foreign materials or bacteria are introduced into the bloodstream (Gravlee et al., 2015).
Arterial Line Filtration Once the CPB circuit is established, the arterial line filter (ALF) becomes a critical component. Positioned between the oxygenator and the patient’s arterial cannula, ALFs are responsible for removing microemboli and air bubbles from the arterial blood before it enters the systemic circulation. The filters typically have a pore size ranging from 21 to 40 microns and can be integrated into the oxygenator itself.
Arterial line filters are crucial because they prevent the entry of potentially harmful embolic material—such as thrombi, fat globules, calcium deposits, and tissue debris—into the patient’s arterial system. Studies have shown that using ALFs significantly reduces the risk of neurological complications, especially stroke, which is a common concern in patients undergoing CPB. However, some research, including a systematic review by Allen et al. (2015), notes that while ALFs are effective in removing microemboli, there is insufficient evidence to conclusively link their use with improved long-term clinical outcomes.
One of the challenges with ALFs is the potential for increased resistance to flow, especially in longer bypass procedures or when the filter becomes clogged. As such, regular monitoring and proper filter selection are essential to maintaining effective filtration without compromising flow dynamics (Gravlee et al., 2015).
Cardioplegia Line Filtration During cardiac surgery, the delivery of cardioplegia (a solution used to arrest the heart) is a crucial step for myocardial protection. The cardioplegia solution, however, is not immune to contamination by microemboli or activated leukocytes, which could contribute to myocardial injury upon reperfusion.
Leukocyte-depleting filters integrated into the cardioplegia delivery system can significantly reduce the number of activated white blood cells and microemboli, thereby minimizing myocardial inflammation and injury. Filters like the Pall LeukoGuard BC1 have demonstrated efficacy in improving cardiac function post-surgery by ensuring that only a cleaner, less inflammatory solution is delivered to the heart (Suzuki et al., 2015).
Furthermore, the importance of cardioplegia filtration is highlighted in studies that show a direct correlation between reduced levels of myocardial enzymes and improved cardiac function following surgery when such filters are employed. These benefits are particularly important in high-risk patients, where even minor myocardial injury could result in significant complications (Gravlee et al., 2015).
Suction and Cardiotomy Reservoir Filtration While there are no dedicated inline filters on suction lines, the cardiotomy reservoir serves as a de facto filter for blood aspirated during surgery. This reservoir captures blood from the surgical field and heart chambers, containing air, fat globules, blood clots, and tissue debris. The reservoir works to filter out larger particles before the blood is returned to the CPB circuit.
The use of the cardiotomy reservoir to filter suctioned blood remains a standard practice. However, there is a growing recognition that blood returned without washing or further filtration can contribute to systemic inflammatory responses and coagulation disorders. This is particularly true in the presence of activated platelets and inflammatory cytokines in suctioned blood. For this reason, many perfusionists prefer to limit the volume of unwashed blood returned to the circuit or use cell-saver devices that allow for the washing of suctioned blood before reinfusion.
Despite its limitations, the cardiotomy reservoir continues to play a vital role in ensuring that larger particles and air bubbles do not enter the CPB circuit, but there remains ongoing research into the benefits of additional filtration steps (Gravlee et al., 2015).
Conclusion Filtration in CPB circuits is essential for ensuring the safety and efficacy of cardiopulmonary bypass procedures. The use of filters at key points in the circuit—arterial line, cardioplegia line, cardiotomy reservoir, and pre-bypass—helps to mitigate the risks of embolism, inflammation, and thrombus formation. As technologies advance, the role of filtration continues to evolve, with improvements in filter design and better understanding of their clinical benefits.
Further research and high-quality clinical trials are needed to define optimal filtration strategies based on patient risk profiles. However, it is clear that effective filtration remains a cornerstone of modern perfusion practices and an indispensable part of cardiac surgery.
References
- Gravlee, G. P., Davis, R. F., Hammon, J. W., & Kussman, B. D. (2015). Cardiopulmonary Bypass and Mechanical Support: Principles and Practice (4th ed.). Lippincott Williams & Wilkins.
- Allen, M. L., Di Tomasso, N., & Nielsen, N. D. (2015). Arterial line filtration during cardiac surgery: a systematic review. Perfusion, 30(8), 635–642. https://pubmed.ncbi.nlm.nih.gov/26607840/
- Suzuki, T., Yoshitake, S., & Suzuki, Y. (2015). Leukocyte filtration in cardioplegia line reduces myocardial injury in cardiac surgery. Journal of Artificial Organs, 18(4), 322–329. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4680653/
- Bojar, R. M. (2001). Manual of Perioperative Care in Adult Cardiac Surgery (4th ed.). Blackwell Publishing.
Asif Mushtaq: Chief Perfusionist at Punjab Institute of Cardiology, Lahore, with 27 years of experience. Passionate about ECMO, perfusion education, and advancing perfusion science internationally.
