Management of Patients with Renal Failure on Cardiopulmonary Bypass
- Home
- Cannulation
- Current Page
Abstract
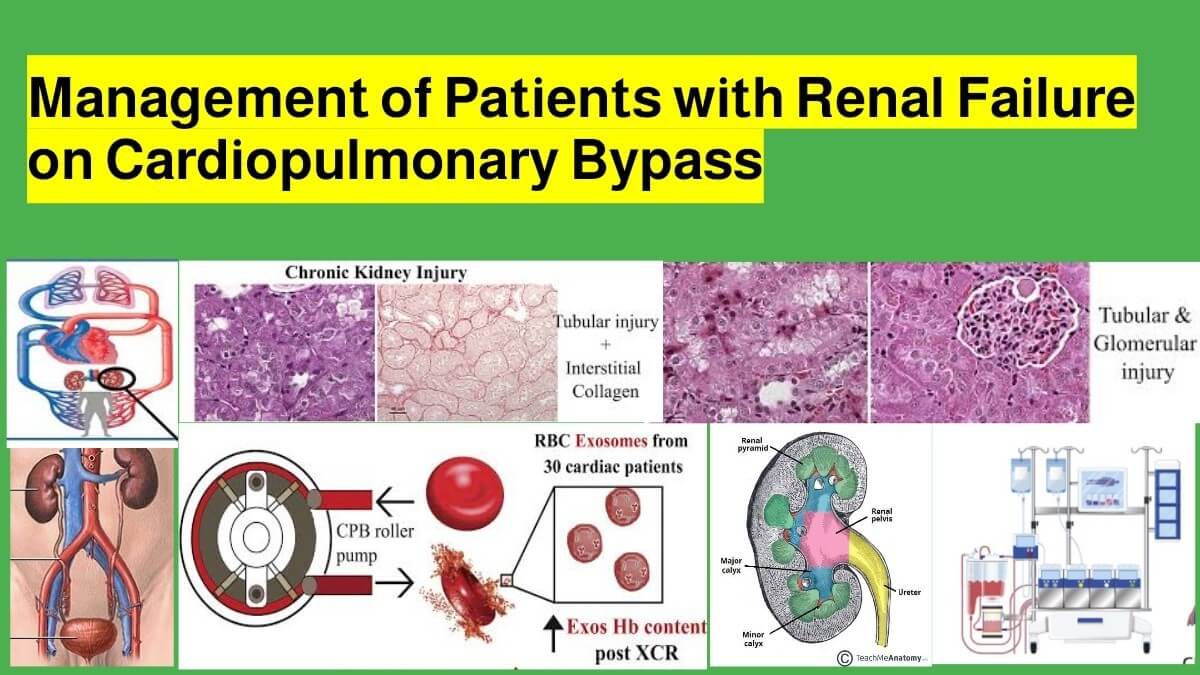
Renal failure presents a significant challenge in patients undergoing cardiopulmonary bypass (CPB), as they are at high risk for acute kidney injury (AKI), electrolyte imbalances, and fluid overload. The management of these patients requires preoperative risk assessment, intraoperative perfusion strategies, and postoperative renal protection measures. This article reviews the pathophysiology of renal dysfunction in CPB, risk stratification, and evidence-based perfusion strategies to optimize renal outcomes in patients with chronic kidney disease (CKD) or end-stage renal disease (ESRD).
1. Introduction
Cardiopulmonary bypass (CPB) is associated with systemic inflammatory responses, hemodynamic alterations, and non-pulsatile perfusion, all of which can contribute to renal dysfunction (Nadim et al., 2016). Patients with pre-existing renal failure have an increased risk of developing CPB-associated acute kidney injury (CSA-AKI), which is linked to higher morbidity, prolonged ICU stay, and increased mortality (Bellomo, Kellum and Ronco, 2012; Thiele et al., 2015).
This article outlines perioperative strategies for managing renal failure in patients undergoing CPB, focusing on perfusion-related interventions to minimize renal complications.
2. Pathophysiology of Renal Dysfunction During CPB
2.1 Hemodynamic Changes
- CPB induces hypotension, non-pulsatile flow, and hemodilution, reducing renal oxygen delivery (Guzzi et al., 2021).
- Loss of autoregulation occurs in patients with CKD, making kidneys more susceptible to ischemia (Schroeder et al., 2020).
2.2 Systemic Inflammatory Response (SIRS)
- Contact activation of blood with CPB circuit surfaces triggers an inflammatory cascade, leading to endothelial dysfunction and renal tubular injury (O’Neal, 2016; Kellum and Lameire, 2018).
2.3 Hemolysis and Free Hemoglobin
- Mechanical trauma from roller or centrifugal pumps leads to hemolysis, releasing free hemoglobin (fHb), which is nephrotoxic and contributes to renal ischemia (Ranucci et al., 2018).
2.4 Oxidative Stress and Reperfusion Injury
- Reperfusion post-CPB leads to oxidative stress, mitochondrial dysfunction, and apoptosis in renal tubular cells (Zarbock et al., 2018).
3. Preoperative Considerations
3.1 Risk Stratification and Optimization
Identifying high-risk patients helps tailor perioperative strategies:
- Preoperative Renal Function Assessment: eGFR, creatinine clearance, and BUN (Nadim et al., 2016).
- AKI Risk Scores: Cleveland Clinic score, KDIGO classification (Mehta et al., 2015).
- Dialysis Patients: Preoperative dialysis is recommended 12–24 hours before surgery to optimize electrolyte and volume status (Lassnigg et al., 2004).
3.2 Medication Adjustments
- Nephrotoxic drugs (NSAIDs, aminoglycosides, contrast agents) should be discontinued (Hoste et al., 2015).
- ACE inhibitors and ARBs may be held preoperatively to maintain renal perfusion pressure (Guzzi et al., 2021).
4. Intraoperative Perfusion Strategies
4.1 CPB Circuit Modifications
- Use of Biocompatible Circuits: Heparin-bonded or phosphorylcholine-coated circuits reduce inflammatory response and platelet activation (Ranucci et al., 2018).
- Miniaturized Extracorporeal Circuits (MiECC) reduce hemodilution and fluid overload (Fromes et al., 2002).
4.2 Perfusion Management
- Mean Arterial Pressure (MAP) Maintenance: Studies suggest MAP ≥ 65 mmHg is optimal for renal perfusion (Nadim et al., 2016; Zarbock et al., 2018).
- Hematocrit Optimization: A target hematocrit of 24–28% is recommended to avoid renal hypoxia (Thiele et al., 2015).
- Flow Rate Adjustment: Cardiac index of 2.2–2.5 L/min/m² helps maintain renal blood flow (Guzzi et al., 2021).
4.3 Ultrafiltration and Hemofiltration
- Modified Ultrafiltration (MUF) helps remove inflammatory mediators and excess fluid, improving renal outcomes (Lassnigg et al., 2004).
4.4 Temperature and Cardioplegia Management
- Mild Hypothermia (32–34°C) reduces metabolic demand while maintaining renal perfusion (Hoste et al., 2015).
- Balanced Electrolyte Cardioplegia is preferred over hyperkalemic solutions in dialysis-dependent patients (Guzzi et al., 2021).
5. Postoperative Renal Protection Strategies
5.1 Early Detection of Acute Kidney Injury (AKI)
- Serum Creatinine Trends: Delayed rise in creatinine is a late marker (Mehta et al., 2015).
- Novel Biomarkers: NGAL, KIM-1, and cystatin C detect AKI before creatinine rise (Zarbock et al., 2018).
5.2 Fluid and Diuretic Management
- Balanced Crystalloids are preferred over normal saline to prevent hyperchloremic acidosis (Hoste et al., 2015).
- Diuretic Use: Furosemide may be used cautiously in oliguric patients but does not prevent AKI (Thiele et al., 2015).
5.3 Renal Replacement Therapy (RRT) Considerations
- Early CRRT Initiation: Indicated in fluid overload, metabolic acidosis, or severe electrolyte imbalances (Bellomo et al., 2012).
- CRRT vs. Intermittent Dialysis: CRRT is preferred for hemodynamically unstable patients (Mehta et al., 2015).
6. Conclusion
Patients with renal failure undergoing CPB require a multidisciplinary approach involving perfusionists, anesthesiologists, nephrologists, and surgeons. By implementing preoperative risk assessment, intraoperative perfusion strategies, and vigilant postoperative renal protection, the incidence of CPB-associated AKI can be significantly reduced.
References
Bellomo, R., Kellum, J.A. and Ronco, C. (2012) ‘Acute kidney injury’, The Lancet, 380(9843), pp. 756-766.
Guzzi, L.M. et al. (2021) ‘Pathophysiology of acute kidney injury in cardiac surgery’, Critical Care, 25(1), p. 108.
Mehta, R.L. et al. (2015) ‘KDIGO clinical practice guidelines for acute kidney injury’, Kidney International Supplements, 2(1), pp. 1-138.
Nadim, M.K. et al. (2016) ‘Cardiac surgery-associated acute kidney injury’, Journal of the American College of Cardiology, 68(14), pp. 1465-1473.
Thiele, R.H. et al. (2015) ‘Perioperative acute kidney injury’, Anesthesia & Analgesia, 120(1), pp. 206-218.
Zarbock, A. et al. (2018) ‘Biomarkers for acute kidney injury’, Critical Care Medicine, 46(1), pp. 86-96
Asif Mushtaq: Chief Perfusionist at Punjab Institute of Cardiology, Lahore, with 27 years of experience. Passionate about ECMO, perfusion education, and advancing perfusion science internationally.
